The electronic configuration of two elements A and B are -
A- 1s2 2s2 2p6 3s2 3p6 4s2
B- 1s2 2s2 2p6 3s2 3p5
What is the formula of the ionic compound formed between these elements?
Step 1: Identify the element
Element A
A has a valence shell configuration of 2. Therefore, A belongs to the Group 2 of the periodic table.
A- 1s2 2s2 2p6 3s2 3p6 4s2
Group 2 elements are metals. Metals have low ionization enthalpy; they can easily lose electrons to attain the nearest stable, inert gas configuration. Group 2 elements can therefore lose two electrons to attain stability.
The principal quantum number of the outermost shell of element A is 4; therefore, the element belongs to the 4th period (row) of the periodic table.
Element A belonging to Group 2 and period 4, is Calcium (Ca).
Element B
B belongs to group 7 based on the valence shell electronic configuration of 7.
B- 1s2 2s2 2p6 3s2 3p5
The Group 7 elements are halogens. The halogens are less by one electron from attaining the stable inert gas configuration. Therefore, halogens have high negative electron gain enthalpy values and attain electrons easily.
The principal quantum number of the outermost valence shell of element B is 3; therefore, the element belongs to the 3rd period (row) of the periodic table.
The element B from the Group 7 and period 3 is Chlorine (Cl).
Step 2: Predicting the Stoichiometry
The Group 2 Calcium (Ca) atom can easily lose two electrons, therefore,
Ca → Ca2+ + 2e-
One Cl atom can only pick one electron; therefore, the two electrons are accepted by 2 Cl atoms to give 2Cl- anions.
2Cl + 2e- → 2Cl-
Net Reaction:
Ca2+ + 2 Cl- → CaCl2
One Ca atom, and two Cl atoms form the compound Calcium Chloride (CaCl2).
Criss-cross (cross-multiplication) method to deduce the Molecular Formula
The simplest method is to employ the criss-cross method.
The valency of Ca is 1, and the one Ca atom comfortably loses two electrons to form Ca 2+ cation.
The valency of Cl is 1, and one Cl atom can accept one electron to form one Cl- anion. However, one Ca2+ cation requires 2 Cl atoms as 2Cl- to balance the charges and form a neutral compound.
By criss-crossing the numerical value of charges and using it as a subscript, the correct ratio of the ions is obtained to conclude the formula of AB2 as CaCl2.
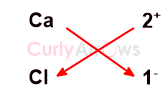
The sign of the charges in the subscript is ignored while writing the molecular formula.
.png)