In substitution reactions, an atom or group of atoms is displaced and replaced by another atom or group of atoms. The displaced atom is called the leaving group. The rest of the molecule remains unaffected.
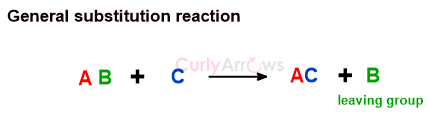
If the substitution reaction goes through a heterolytic cleavage mechanism, then depending on the reagent, the reaction is of two types- nucleophilic and electrophilic substitution. The substitution reaction is free radical if the reaction mechanism goes via homolytic bond cleavage.
a) Nucleophilic substitution reaction (reagent nucleophile + leaving group nucleofuge)
It is the reagent that carries an extra set of electrons and acts as a nucleophile in a reaction with the substrate. Therefore, in the reaction, the nucleophile provides the bonding electrons to form a new bond with the substrate.

After bonding with the nucleophile, the leaving group comes away with an electron pair. The leaving group is also called a nucleofuge. The breaking away of the leaving group maintains the valency of the atom.
Therefore, the heterolytic bond cleavage, nucleophile, and nucleofuge determine a nucleophilic substitution reaction.
The substrate nature further determines the subtypes of the nucleophilic substitution reaction.

Aromatic and aliphatic halides undergo nucleophilic substitution reaction by this mechanism where the leaving group is a halogen, especially Chlorine and Bromine.
b) Electrophilic substitution reaction (reagent electrophile and leaving group electrofuge)
The reagent is an electrophile in electrophilic substitution reactions, especially in benzenes. Aromatic compounds like benzenes undergo electrophilic aromatic substitutions, and the leaving group is an electrofuge, H+.

The substitution reactions on the benzene ring allow for the introduction of many functional groups and the multi-step synthesis of various products.

c) Free Radical Substitution
The free radicals are produced by homolysis in a previous step that reacts with the substrate and displaces an atom. The displaced atom is also a free radical that goes on to react further. The free radical formation and the reaction always occur through homolytic bond cleavage.

The homolytic bond fission in enabled in the presence of heat or light.
The Hunsdiecker and Wohl–Ziegler reaction are examples of free radical substitution reactions.
