Dalton, in 1804, in his work on ‘Atomic Theory,’ proposed the law of chemical combination to explain how atoms form compounds. According to him, atoms of different elements combine in a simple whole-number ratio to give compounds.
For example, two Hydrogen and one Oxygen combine to form water (H2O), or four Hydrogens and one Carbon combine to form methane (CH4), or one Nitrogen combines with three Hydrogens to give ammonia (NH3).
While the theory simplified the process of formation of compounds, it did not provide any information on How they form such compounds. What Characteristics must an atom have to form compounds? What forces bring the atoms together to form a compound? What forces are responsible for the atoms to remain stuck in a compound?
While the early scientists in the 1900s understood that something holds atoms together in the molecules, in their undergraduate years, they only studied it as ‘the hook and eye mechanism’ to explain bonding between atoms. Each atom had hooks (?)to attach to other atoms and eyes (O) so that other atoms attached to it to form long chains of covalent bonds.
In 1916, Lewis tried to explain bonding by proposing the structure of an atom as a cube. According to him, the outermost electrons are the only ones responsible for bonding, an idea he picked up from Langmuir.
Lewis and Langmuir believed that the most stable arrangement of the outer electron is eight. Lewis postulated that the symmetrical arrangement of eight electrons is only found in a cube such that the electrons are farthest apart. He called the kernel the central positive nucleus; meanwhile, he avoided showing other electrons in the cube since they were not involved in bonding.

The atoms combined to form bonds by adding and replacing the electrons in the cube. So, if an atom only had seven electrons in its outer shell, it would require eight for stability. It would try to combine with another atom with an extra electron, and the two atoms will try to share an electron to form a stable product. This shared electron bond is what tied atoms together into a molecule. For Lewis, the shared electron bond was the side of the cube that held the two atoms together.
The cube structure of an atom could explain how a carbon atom can form bonds with other elements while keeping its tetrahedral shape. But it could not explain the formation of triple bonds and free the rotations of the single bonds. It could not describe the shapes of many compounds.
Eventually, the cubical atom was discarded for Neil Bohr’s structure of an atom in the year 1913.
Breakthroughs were happening in the field of physical chemistry in the 1920s with physicists, mathematicians, and chemists- Heisenberg, Schrödinger, Heitler, London, Pauling, Einstein, Planck, etc. trying to resolve what an atom looks like and how it bonds to form molecules giving rise to a field of quantum mechanics.
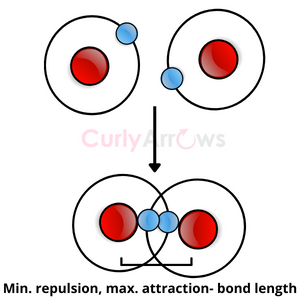
In 1927, Heitler and London tried explaining the formation of the simplest Hydrogen molecule (H2). They postulated that the atoms of Hydrogen approach when there is an attraction between the electrons and the nucleus until the electrons reach so close that they become part of both atoms’ nuclei, continuously exchanging positions. This attraction is balanced by the repulsion of the two positively charged nuclei creating a chemical bond of definite length and strength. The electrons are concentrated between the two atoms in the bond.
The Heitler-London method formed the basis of the Valence Bond Theory. The Valence Bond Theory could not prove the tetravalency of Carbon and geometries of other molecules.
In the years following 1924, scientists believed the electron to possess both the wave and the particle behavior. Attempts also began to assign specific positions to the electrons around the nucleus. The regions wherein the probability of finding an electron is the highest were called the orbitals. These orbitals were arranged in the ascending order from the closest to the farthest from the nucleus. The orbital energies also increased in that order.
Linus Pauling, in 1927, came up with a model of covalent bond formation wherein he proposed the mixing of an atom’s orbitals to form a new set of molecular orbitals with a process known as Hybridization. The electrons, after mixing, occupy new positions in the molecular orbitals, with the chance of covalent bonding being highest if the low-energy orbitals are first occupied.
The electrons in the molecular orbitals are spread out at specific bond angles to give new shapes to the molecules. This avoids any repulsion arising from the bond electrons. The covalent bonds thus formed in the molecule are equal in energy.
The hybridization theory could explain the formation and shapes of polyatomic molecules. Future advances in understanding covalent bond formation are covered under Molecular Orbital Theory.
