Different arrow notations are frequently encountered in Chemistry, mainly Organic Chemistry. Each one has a specific purpose and cannot be used interchangeably.
A few of the most common ones are:
a) Chemical Reaction Arrow
The conversion of a reactant to a product is commonly shown with a chemical reaction arrow. Most of the fundamental reactions in chemistry (addition, substitution, displacement, decomposition, etc.) are expressed in a chemical equation using this reaction arrow.
The arrow's tail lies towards the substrate. The head points to the products, implying that the reaction always proceeds to product generation. Therefore, the arrow is also called the forward arrow since it shows the direction in which the chemical reaction proceeds.
The reagents and the catalysts are written above the arrow, whereas the reaction conditions (time, temperature, solvent) are below.
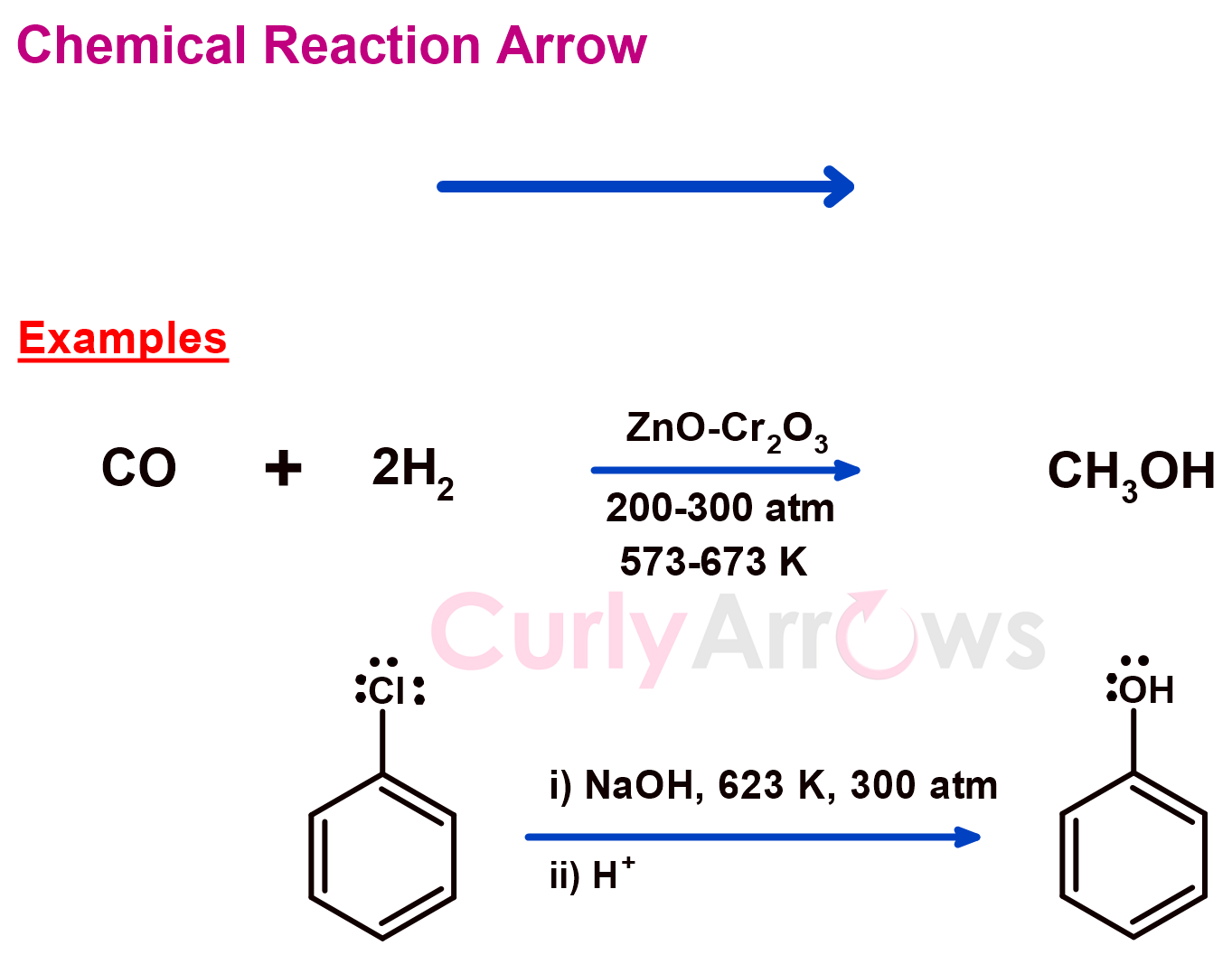
If the product formation involves more than one reaction, in that case, a single reaction arrow can be used to denote all the required steps. The subsequent reactions are numbered, mentioning the reagents and the reaction conditions. They are written above or below the chemical reaction arrow.
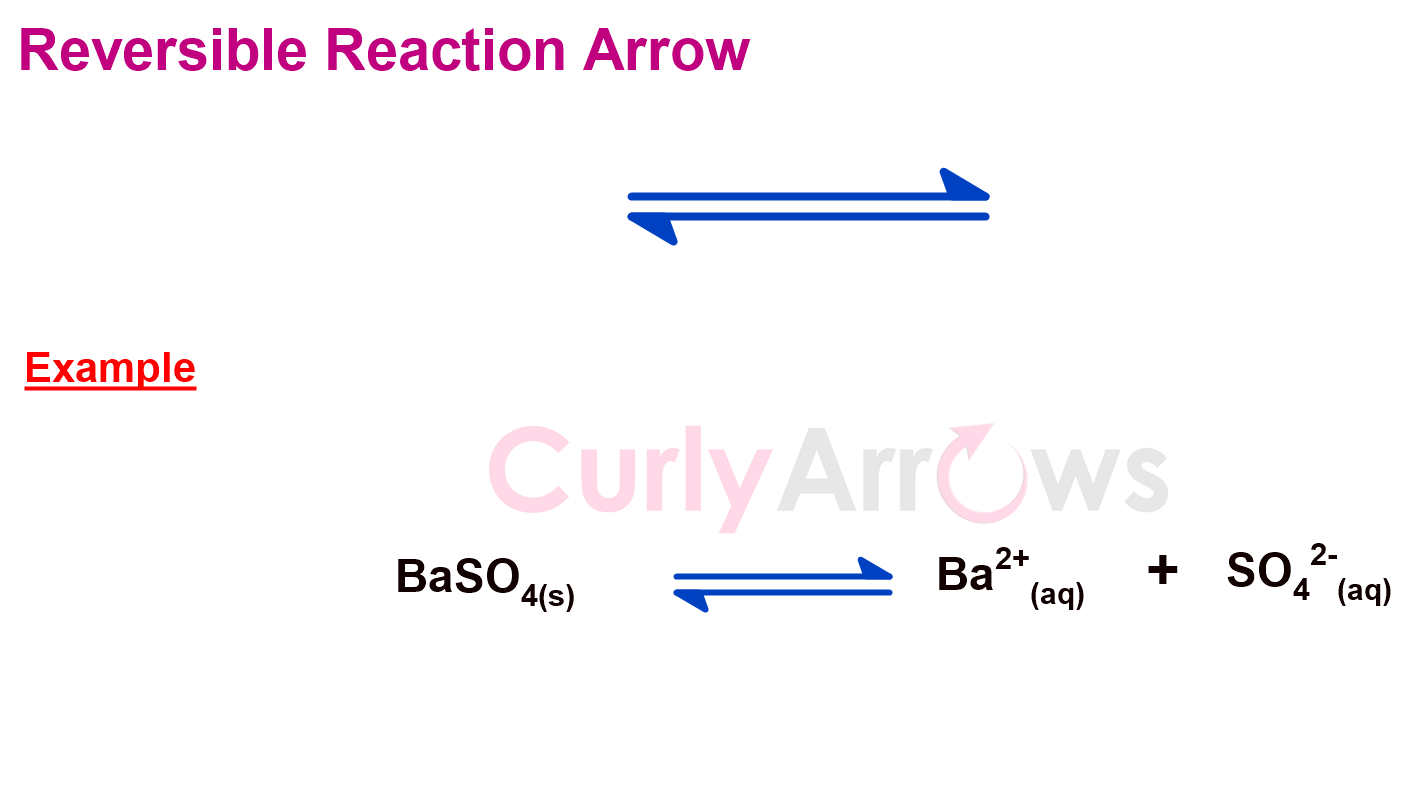
b) Reversible Reaction Arrow
In a typical chemical reaction, the reactants always proceed in the forward direction to form the products. However, in the case of the reversible reaction, the backward or the reverse reaction also occurs. The products formed tend to react to give back the starting material. Both the reactions co-occur, and there is no complete consumption of the reactants or the products.
A reversible reaction arrow is, therefore, bidirectional, and half-headed. The tail end of a reversible reaction arrow lies near the reactants, and the arrowhead always points towards the products.
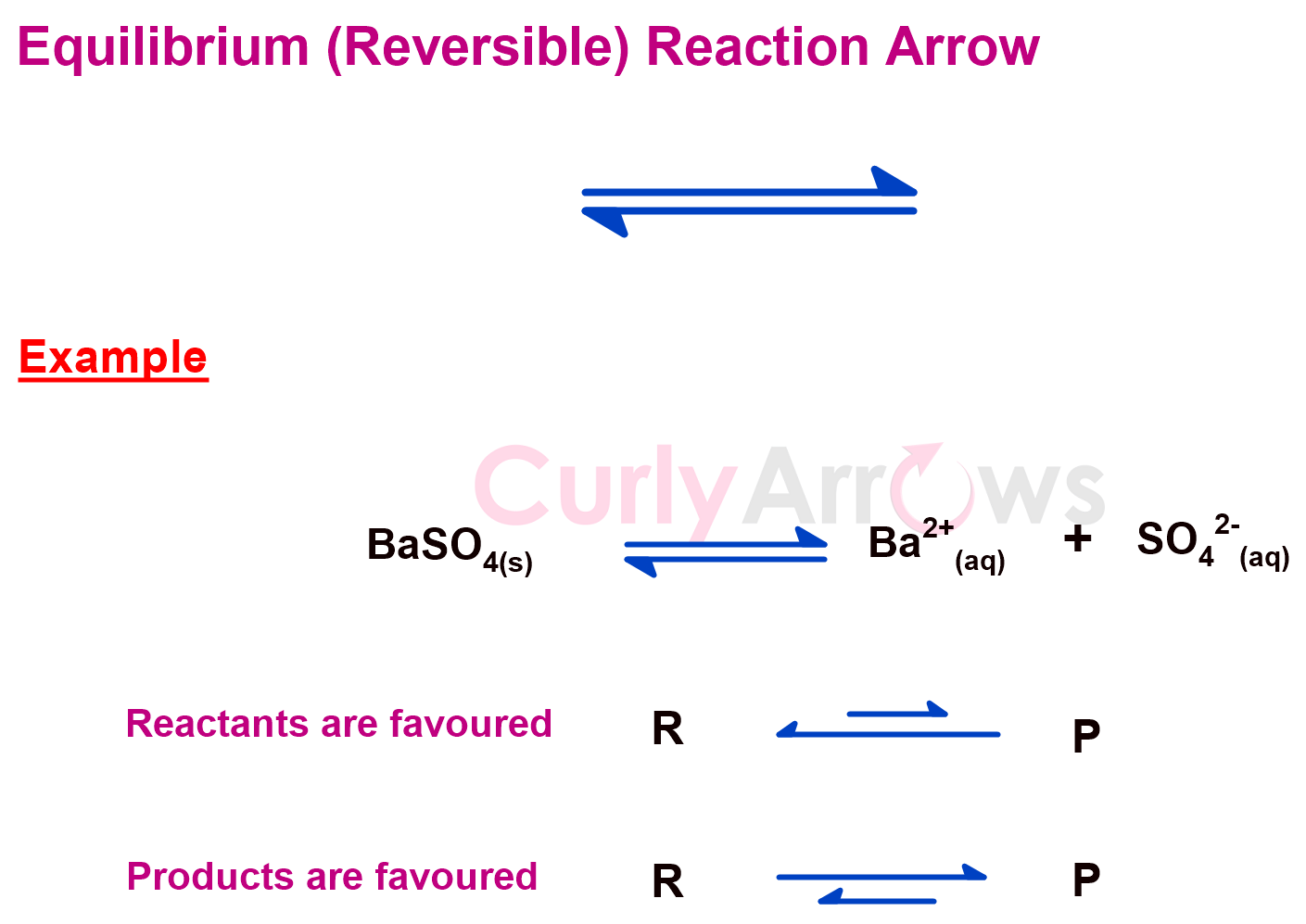
c) Equilibrium Reaction Arrow
In a reversible reaction with similar rates, the forward and the reverse reaction leads to an equilibrium, a state of chemical reaction that does not go till completion. Therefore, the longer reaction time does not cause any significant progress and does not lead to any change in the reactant's concentration or product.
Suppose the concentration of both the reactant and the product are equal. In that case, the equilibrium reaction arrow looks like the reversible reaction arrow. The arrow length is the same.
However, sometimes, altering the reaction conditions can change the direction of the reaction favouring the formation of the reactants or the products.
A longer forward reaction arrow indicates that the forward reaction occurs more than the reverse reaction. However, when the reverse reaction is dominant, the length of the arrow is longer.
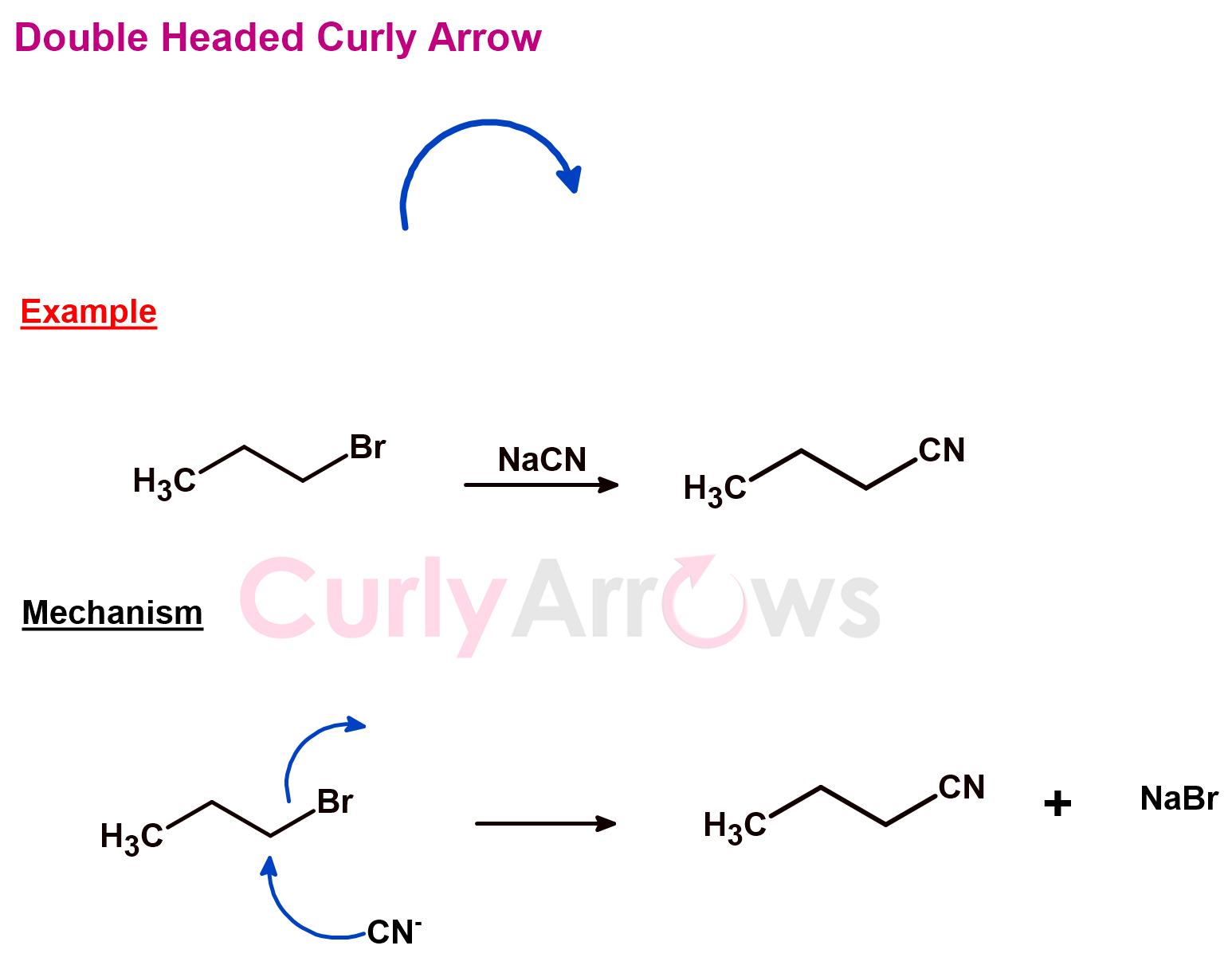
d) Double Headed Curly Arrow
In chemistry, the transformation of organic molecules occurs by the loss or the gain of electrons. These changes on the paper are indicated using the curly arrows, which show electrons’ path from one point to another; essentially, two electrons’ movement. The curly arrows have a spearhead (double-headed). The tail end of the arrow is at the electron-rich atom (called the nucleophilic center) and the arrowhead on the electron-deficient atom (known as the electrophilic center).
e) Fish-Hook Curly Arrow
While the curly arrow shows two-electron movement, the movement of one electron is shown using the fishhook curly arrow. This single electron transfer reaction is also known as the free radical reaction. Unlike the curly arrow, it has a half-head (half of a spearhead) and looks like a fishhook.
The tail end is at the electron and the arrowhead on the atom that will possess the radical/electron. The fishhook arrow mostly appears in pairs to account for all the electrons.
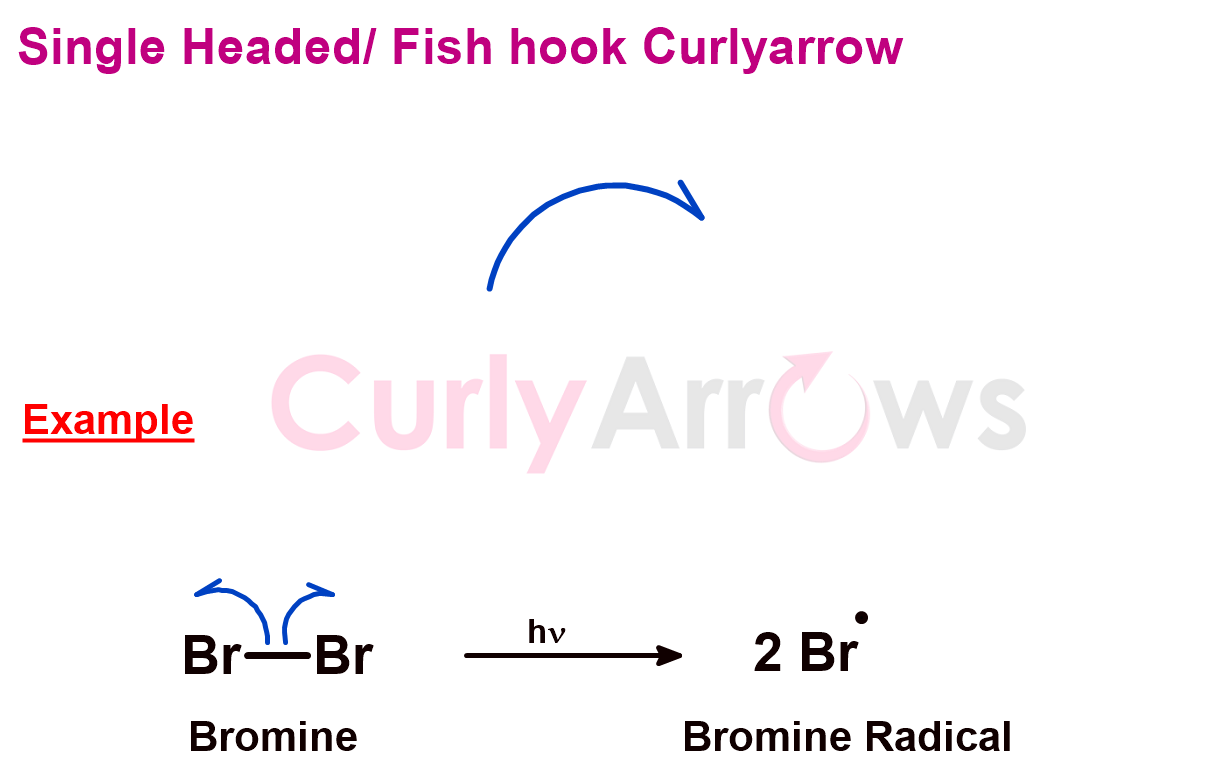
For example, a homonuclear diatomic molecule, Br2, has a two-electron covalent bond. The electrons are shared equally between the two atoms. However, each electron resides on one Br atom to generate two free Br. radicals on the homolytic bond cleavage. This electron transfer is shown by using the fishhook arrow.
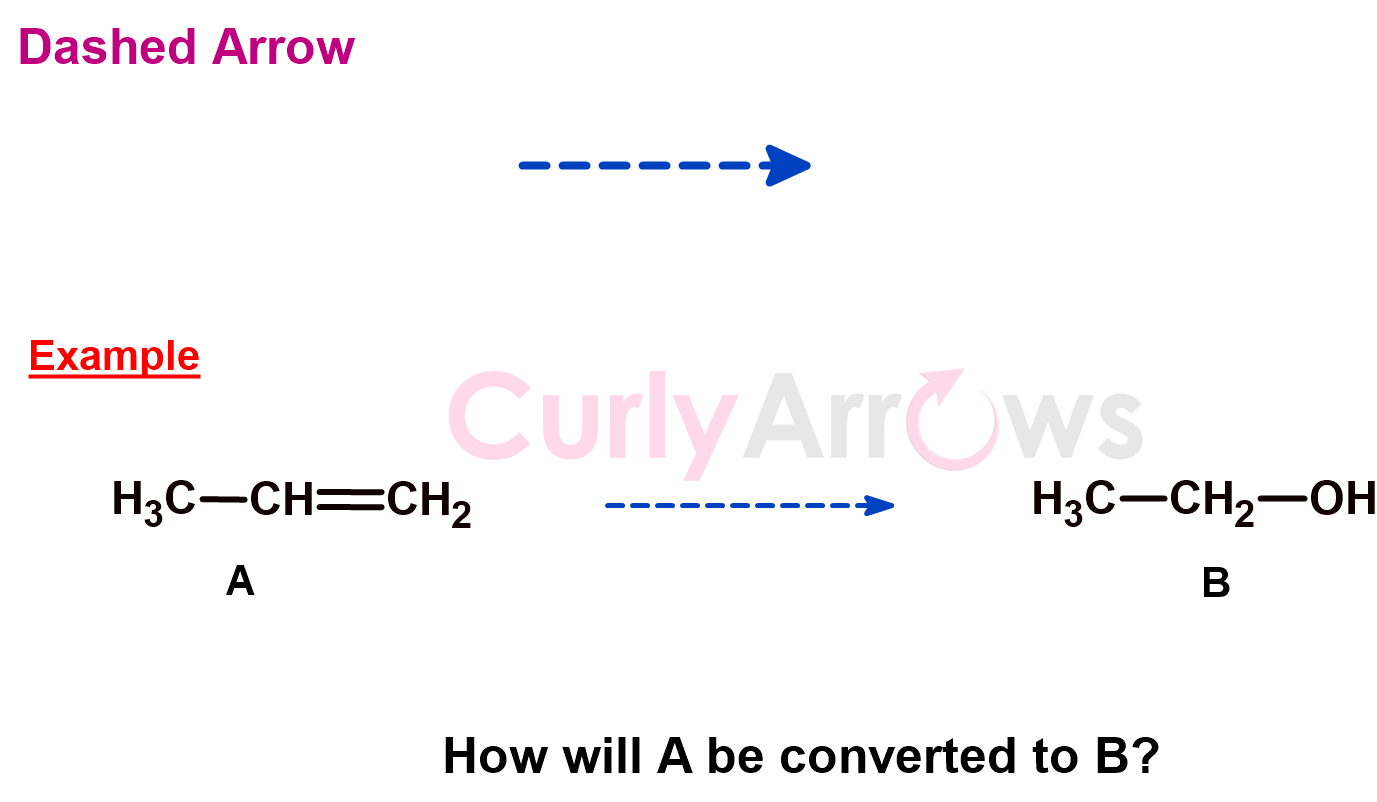
f) Dashed Arrow
A reaction to convert a substrate to a product is proposed using a dashed arrow. The reaction must be experimentally verified and confirmed. If the reaction was successful, the arrow later changes from dashed to the chemical reaction arrow.
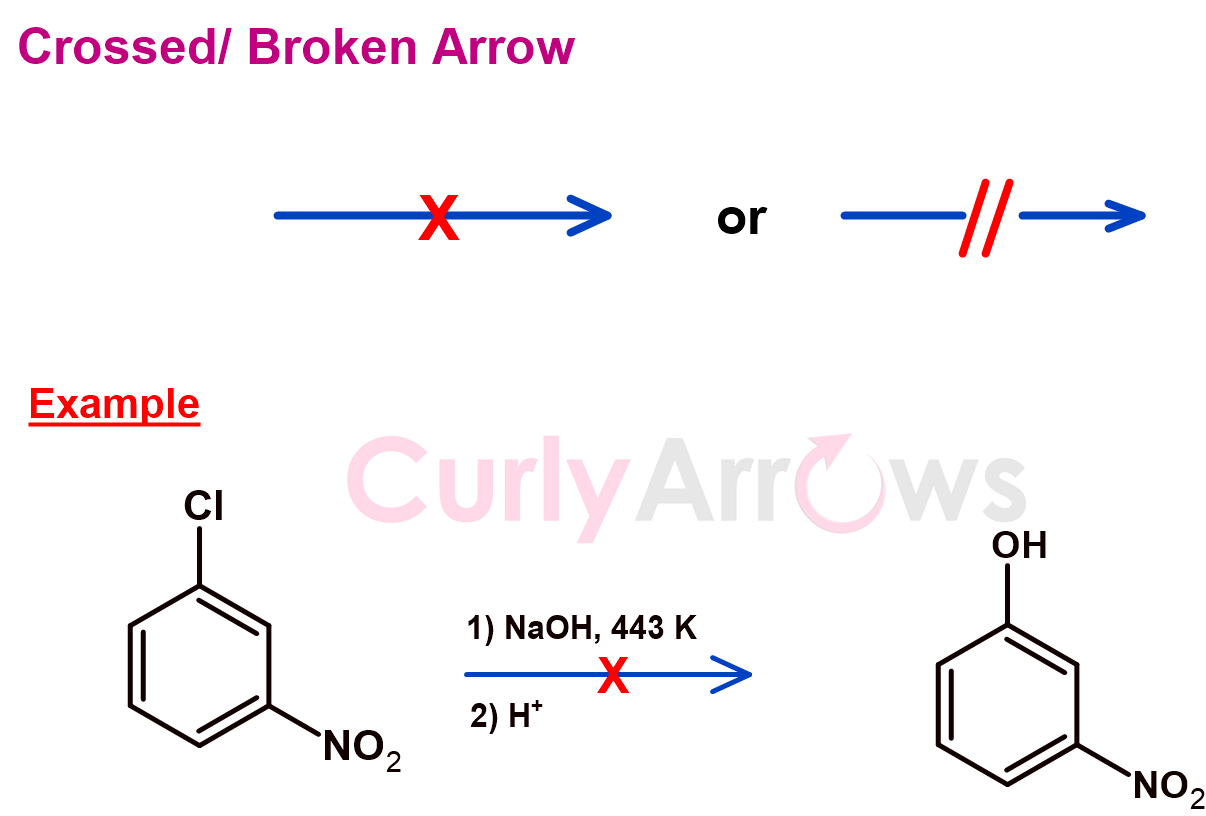
g) The Crossed or The Broken Arrow
The crossed arrow indicates a failed chemical reaction; the reaction did not yield the desired product/s. Therefore, a cross sign is drawn on the reaction arrow, or sometimes it is shown as broken.
h) Resonance Arrow
Some molecules' electronic structure (Lewis structure) can be drawn in more than one way. Each proposed structure can explain some of the observed molecular properties. Not one structure could explain all the observed properties.
The electron delocalization in the molecule is thought to contribute to the behavior, and the phenomenon is called resonance.
The path of electron delocalization is shown with a set of structures, called the contributing/resonance structures. A two-headed resonance arrow separates each structure.
The actual molecule possesses the characteristics of all the contributing structures and, therefore, is called a resonance hybrid. The electron movement/delocalization is shown with the curly arrows in the individual resonance structures. The electrons move in a clockwise manner. The position from which the electrons are delocalized acquires a positive charge. Furthermore, the atom that acquired the electrons is shown with a negative charge.
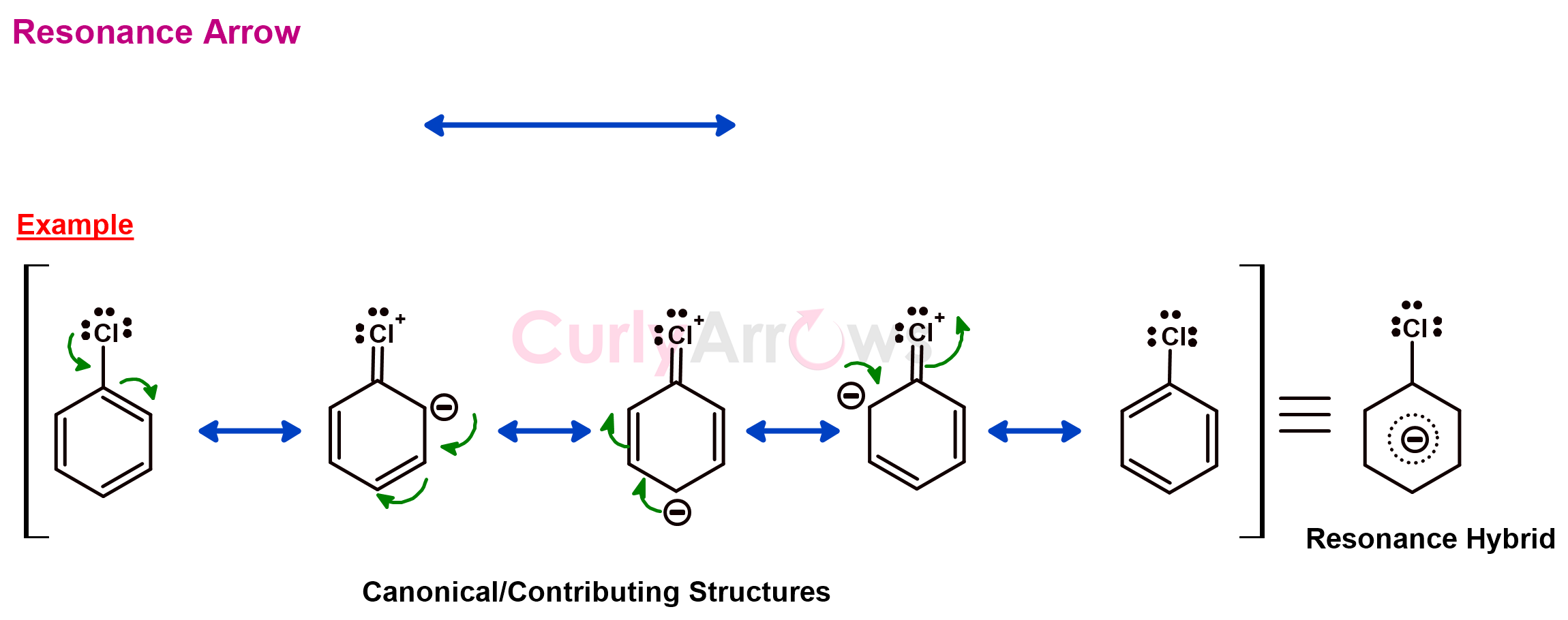
Nowadays, the term delocalization is used instead of resonance to avoid confusion. The structures are now separated by a comma instead of a double-headed resonance arrow.
i) Retrosynthesis Arrow
The planning of the chemical synthesis of a target molecule is done using the retrosynthesis arrow. The arrow is made of two straight lines and has one single head.
The arrow informs that the product on the left (tail end) is made using the starting materials on the right (facing the arrowhead).
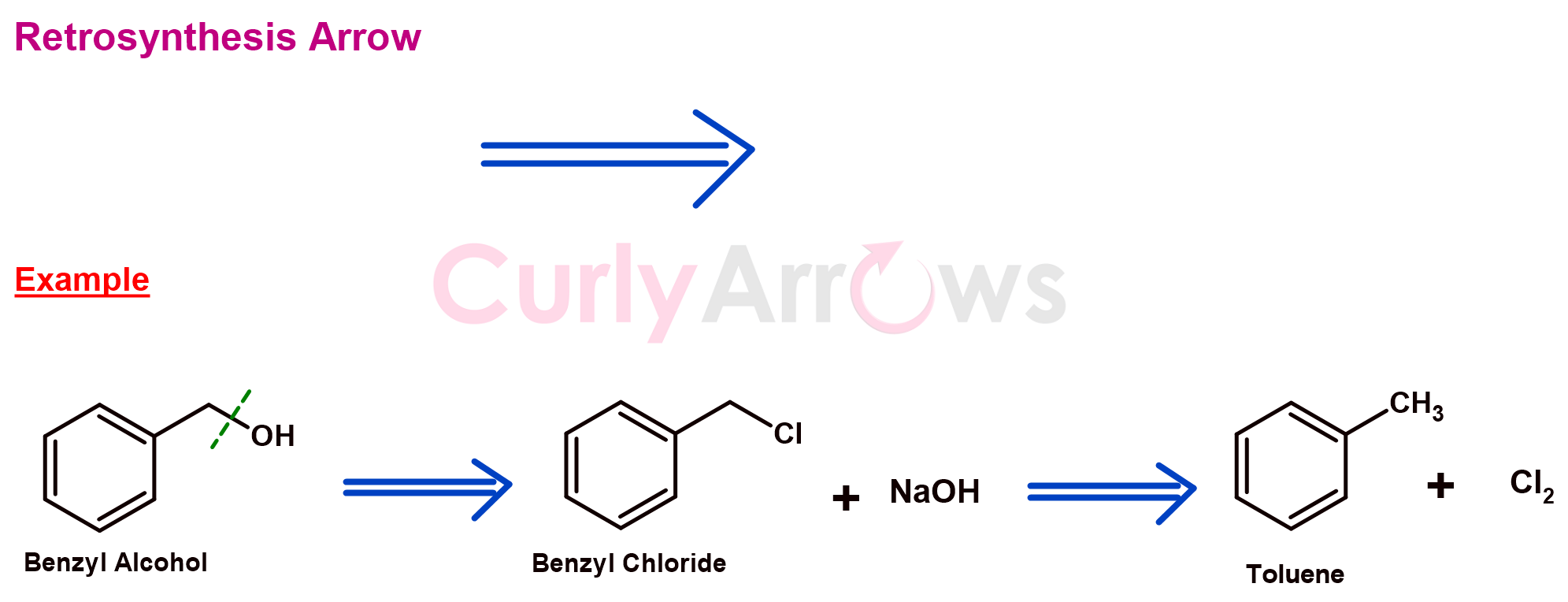
In retrosynthesis planning, the product is the starting point. The product molecule is broken down into smaller fragments stepwise to arrive at simpler starting materials. The fragmentation uses chemistry rules, reasoning, and understanding chemical reactions.
j) Rearrangement Reaction arrow
A rearrangement reaction arrow looks like a chemical reaction arrow but has a middle loop. The arrow is used to show the rearrangement reaction in one single step. However, the actual mechanism of the rearrangement reaction is shown in several steps using the curly arrows and the chemical reaction arrows.
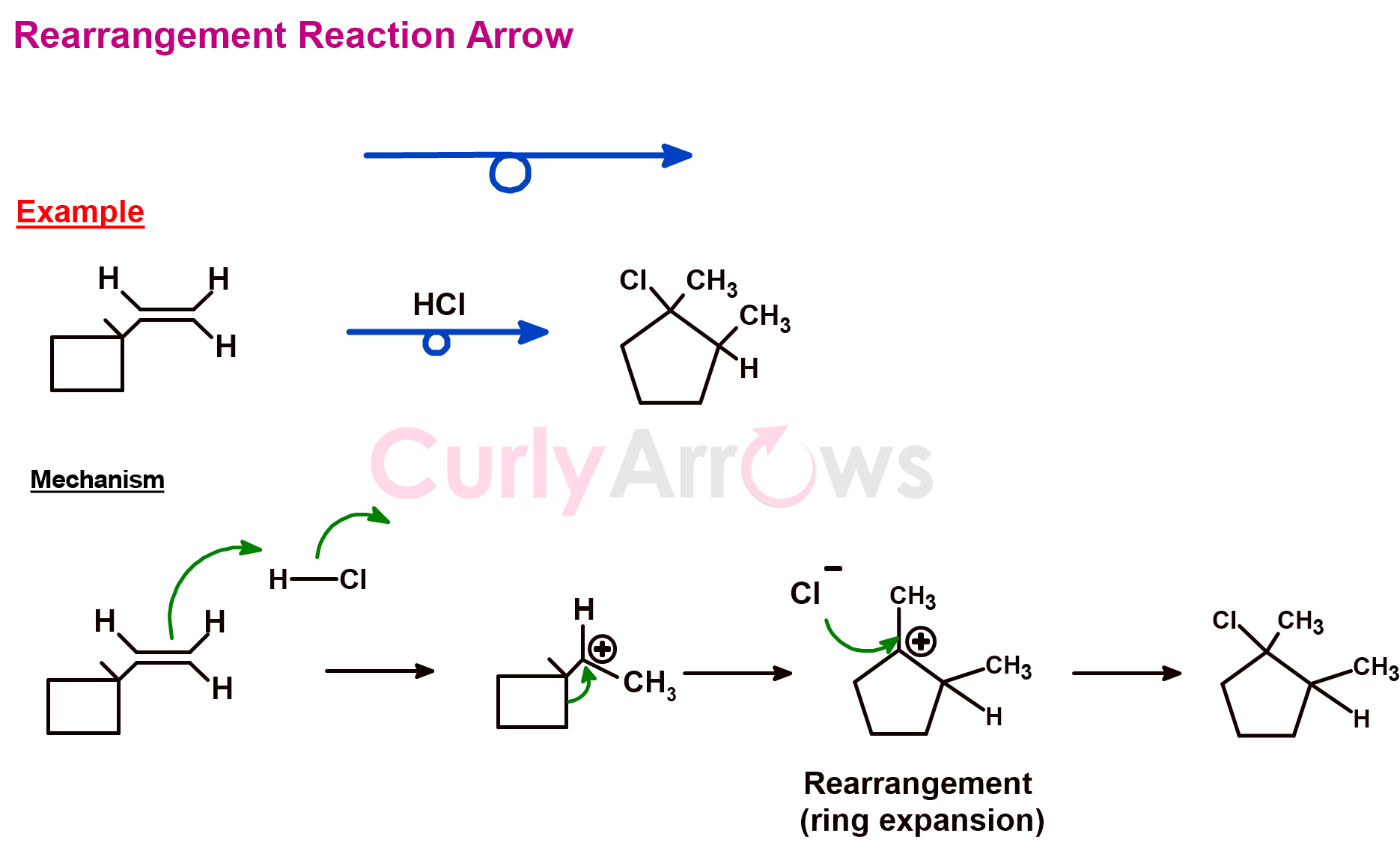
The rearrangement reactions may involve the breaking of several bonds. Also, the rearrangements may occur within the molecule (intramolecular) or between two molecules (intermolecular).
Other Arrow Notations
1) Reflux Reaction Arrow
A solvent used in a chemical reaction is boiled at its boiling point that can convert the liquid to vapor, causing evaporation. However, using a water condenser, these hot vapors can be cooled and condensed back to the liquid. Hence, the solvent volume remains constant, and there is no loss due to evaporation. Such a laboratory technique is known as reflux.
A reaction that requires a refluxing condition is denoted using two full-headed arrows pointing upward and downward. The arrows are written below the reaction arrow, usually next to the solvent.
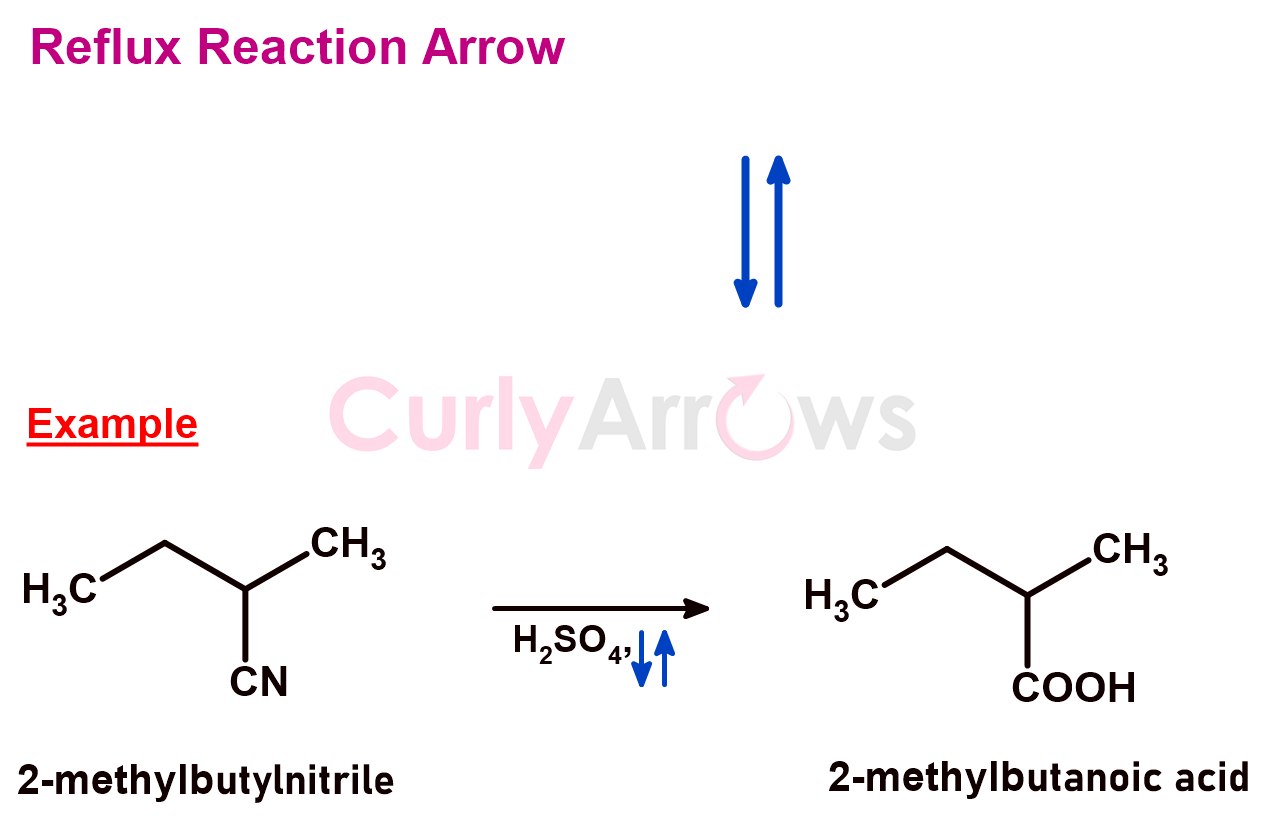
2) Orbital Electrons Arrow
While writing the electronic configuration of an atom, the position of the electrons in the shell (1, 2, 3, etc.) and a sub-shell (s, p, d, f, etc.) is indicated by using the upward or downward arrow. One electron is one arrow, and the arrows may be drawn as a half-head or a full head.
When two electrons are part of the same sub-shell, they have opposite spins. Both the upward and the downward arrows are drawn side by side—for example, s-orbital electrons.
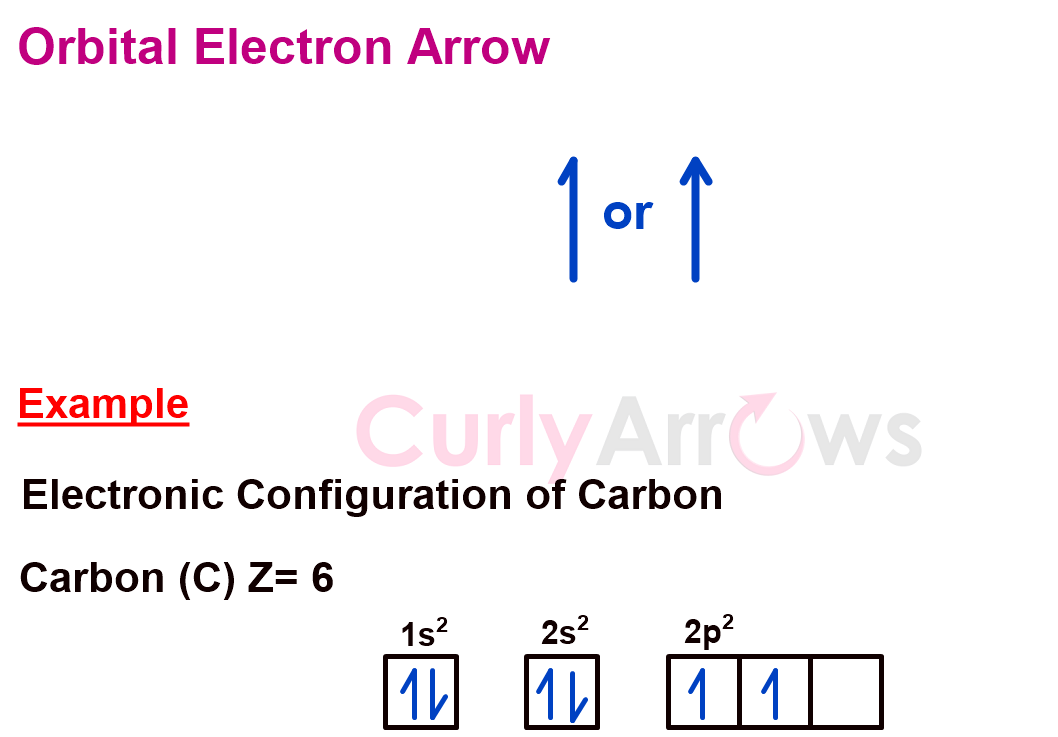
The arrows also appear in molecular orbital energy level diagrams to show the electron position.
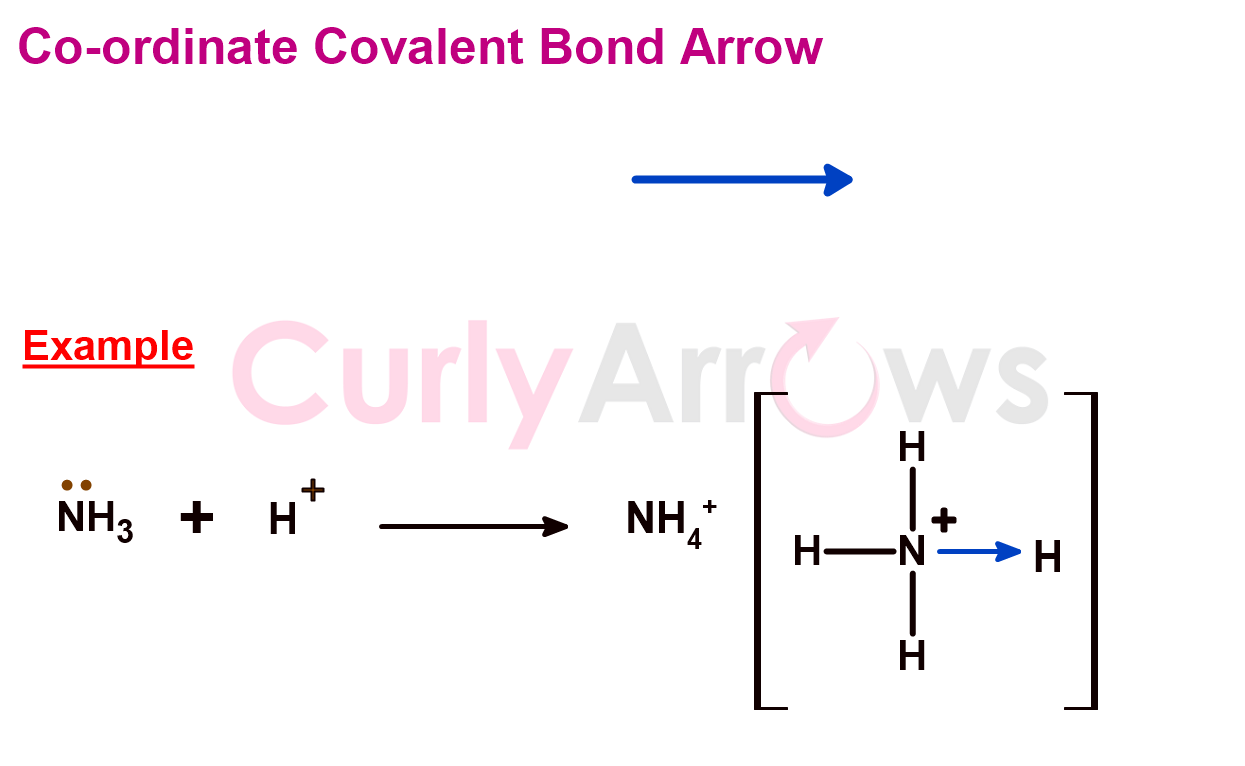
3) Co-ordinate Covalent Bond Arrow
Suppose in a two-atom bond; one atom is the donor and the other an acceptor, then, in that case, the bond is called a coordinate covalent bond. The bond between the two atoms is indicated using an arrow. The arrow's base points to the electron-rich atom (the donor). The arrowhead faces the electron-deficient atom that accepts the electron pair.
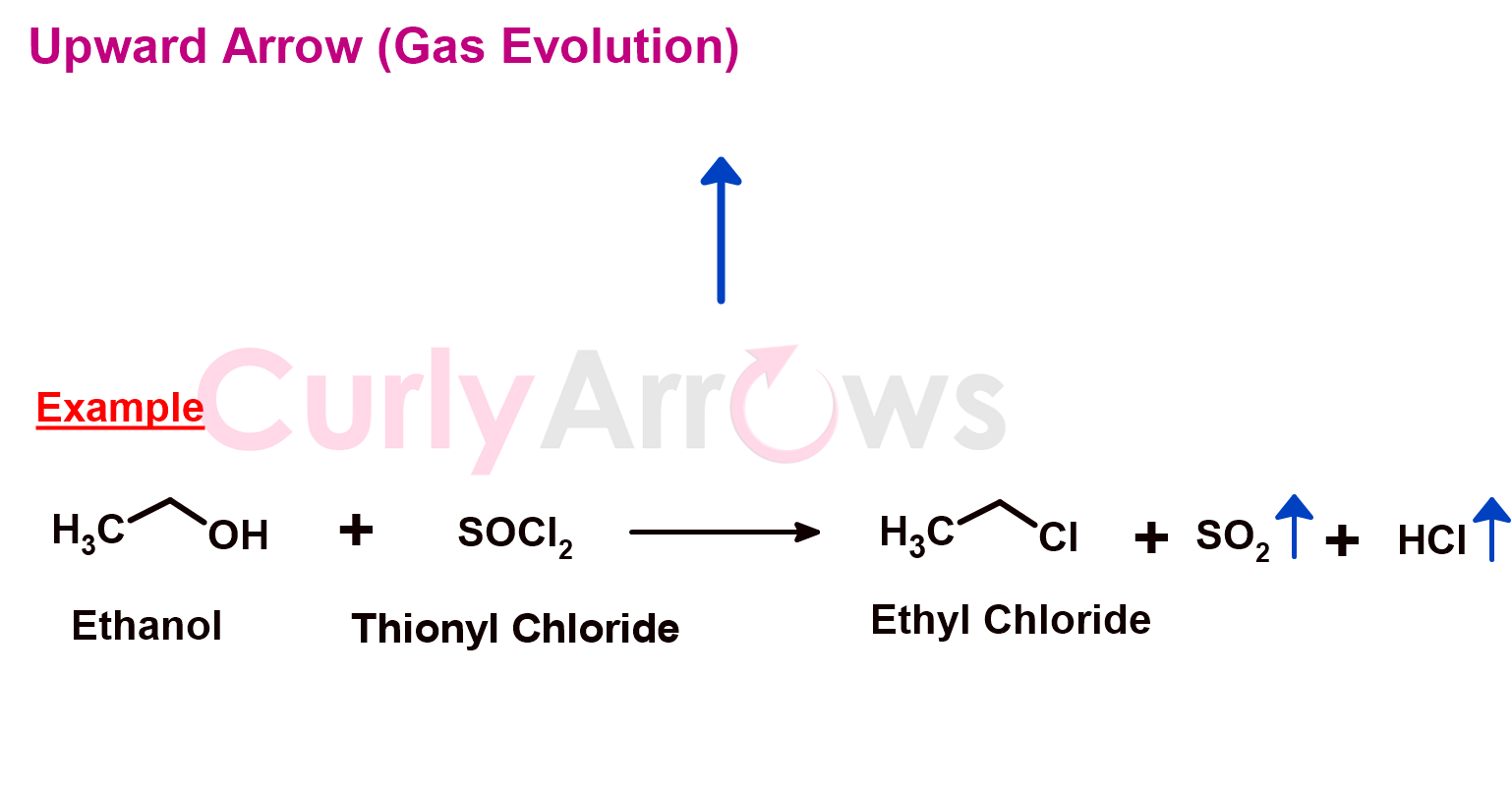
4) Upward Arrow (Gas Evolution)
Suppose any chemical reaction product is gas, then it tends to escape from the reaction mixture. In that case, it is indicated by an upward-facing spearhead arrow. It is drawn next to the gaseous product on the right-hand side of the chemical reaction equation.
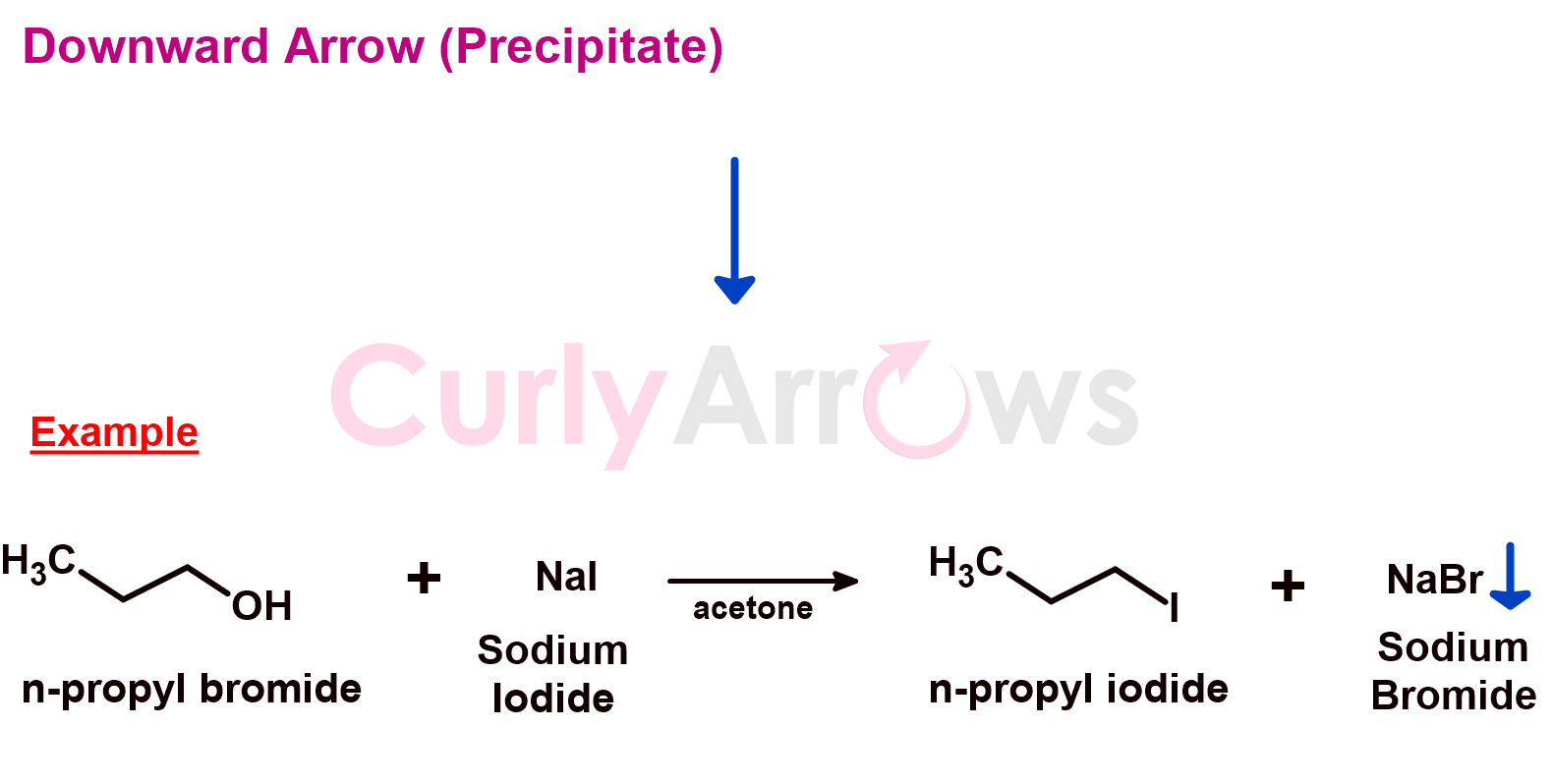
5) Downward Arrow (Precipitate)
Suppose one of the products of a chemical reaction is an insoluble precipitate. In that case, its formation and deposition are indicated using a downward-facing spearhead arrow. It is written on the right-hand side next to the precipitated product in the chemical reaction equation.
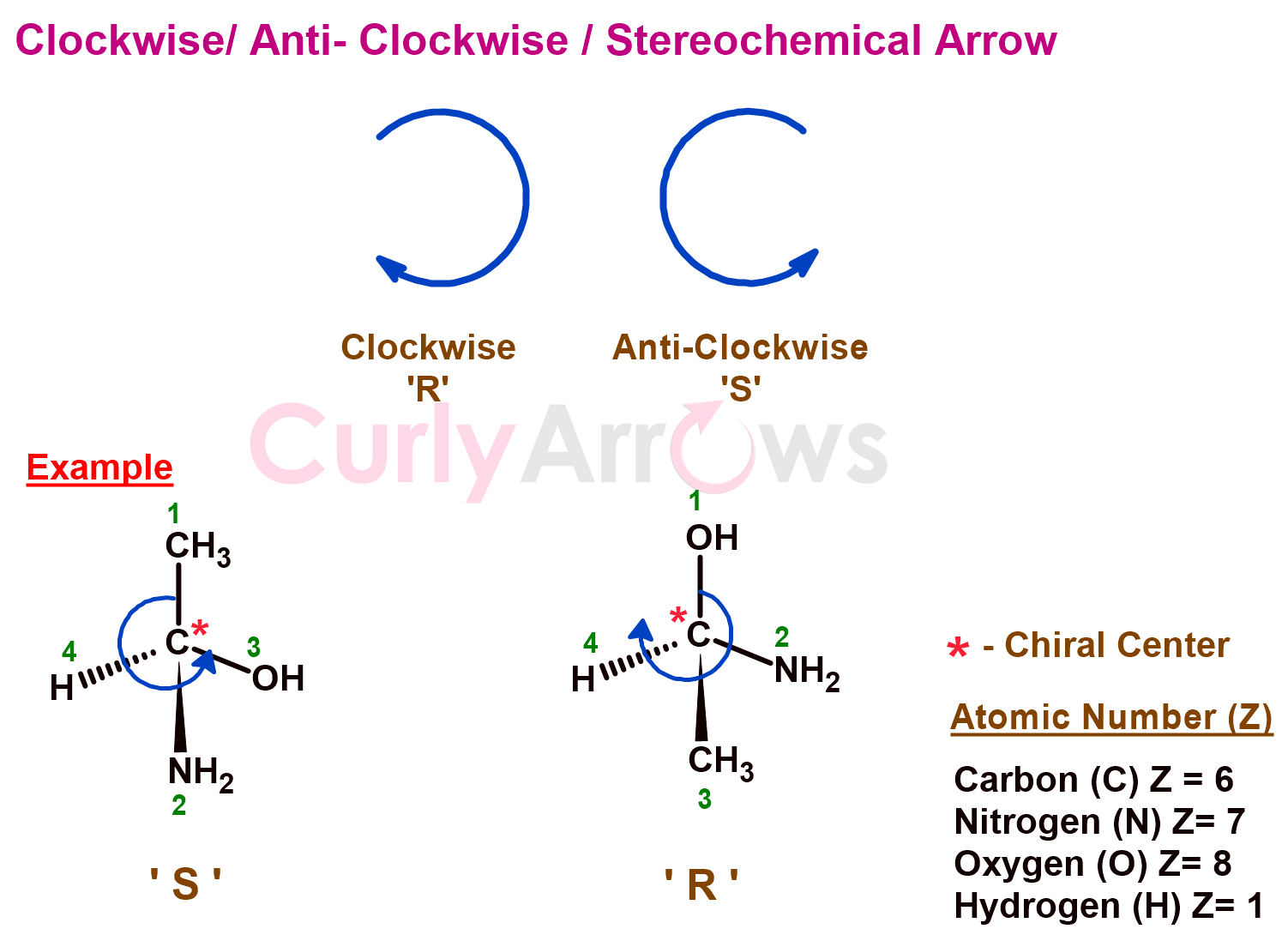
6) Clockwise and Anti-Clockwise/ Stereochemical Arrow
A spearhead curly arrow indicates the molecule's absolute configuration, which is related to the spatial arrangement of atoms around a stereogenic centre. A stereogenic centre is also called a chiral centre, an atom attached to four different groups.
Based on the Cahn-Ingold-Prelog naming system, based on the atomic number (Z), some groups prioritize others. Higher the atomic number, the higher the priority. These groups are numbered, and a curly arrow is drawn based on whether the groups are arranged clockwise or anti-clockwise.
If the rotation of the arrow is clockwise, in that case, the configuration is labeled 'R' (rectus/clockwise). If the arrow rotates anti-clockwise, the configuration is labeled 'S' (sinister/anti-clockwise).
7) Wavy Arrow
The wavy arrow represents a photon of light. It shows an energy emission or an energy transfer.
The wavy arrow expresses the wave-particle duality of light and electron. The ‘wave’ nature of the photon is shown as the long wiggling tail of the arrow, and the ‘particle’ part is the arrowhead carrying one energy unit.
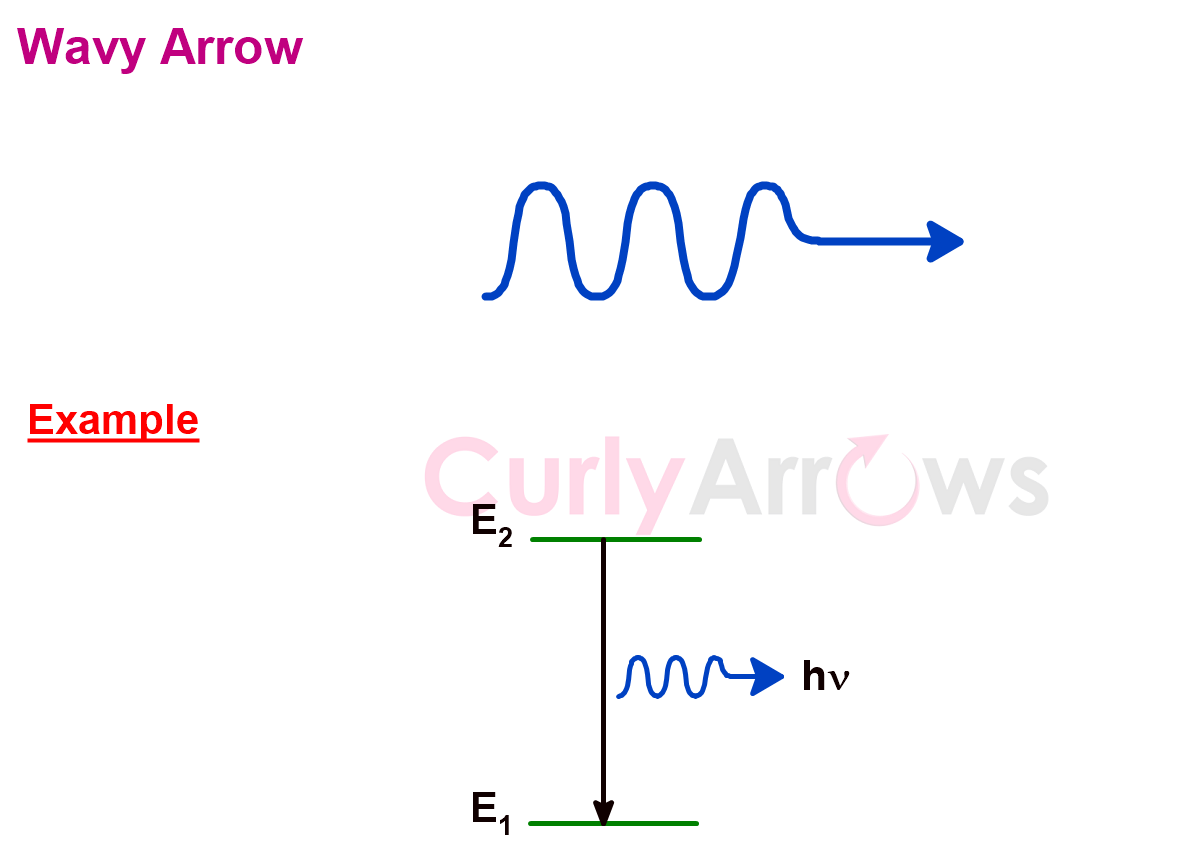
When the arrow is drawn between two energy levels, it shows the transition from a higher energy state to a lower energy state by photon emission.
In photochemistry, the wavy arrows represent a non-radioactive decay.
8) Dipole Moment Arrow
Suppose two atoms in a covalent bond have an electronegativity difference in the range of 0.5-2; in that case, a dipole is induced in the molecule. The more electronegative atom pulls the electron density in the bond towards itself, thereby carrying a partial negative charge (δ-). The less electronegative atom gets a partial positive charge (δ+). Therefore, the dipole is a directional vector quantity.
The bond dipole arrow is drawn parallel to the covalent bond. The arrowhead points to the electronegative atom. The tail end has a built-in + sign that faces the less electronegative atom, the positive terminal of the dipole.
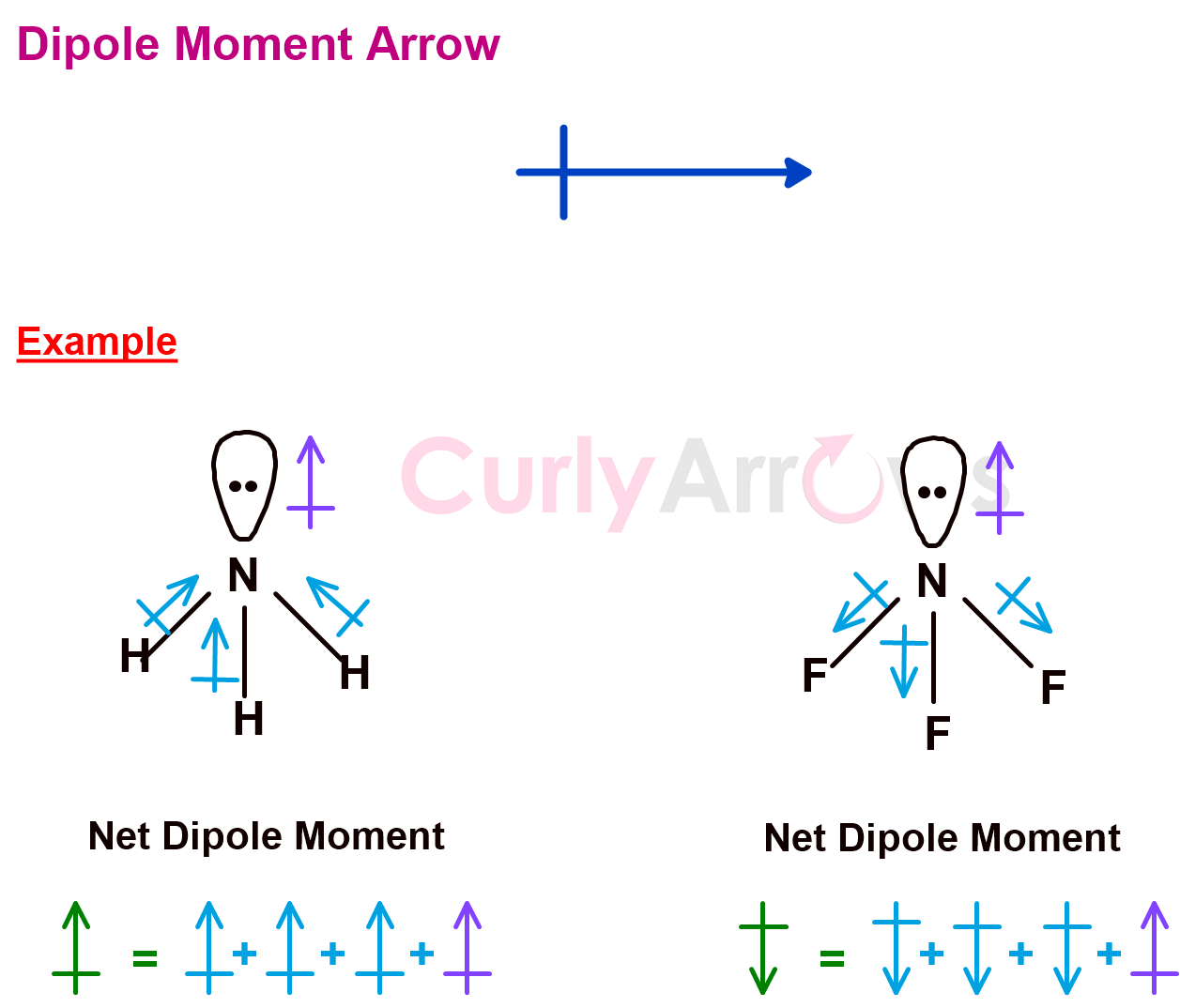
The resultant dipole moment of the molecule, which is a sum of the bond dipole moments, is also shown using the same arrow. If all the individual bond moments add up, the resultant Dipole Moment is higher. If a few of the individual bond moment cancels out, the resultant Dipole moment of the molecule is lower.
Take the Quiz
Quiz on Curly arrows Basic Concepts
