Once each electron of the two atoms forms the covalent bond, the electrons are simultaneously distributed between them so that they belong to both the atom’s nuclei. This area between the atoms is called the region of electron density.

The two electrons of the bond can be shared equally or unequally between the two atoms. Based on how the electrons in the covalent bond (bond pair) are distributed, the covalent bond is divided into two types- polar and nonpolar.
The electron distribution is equal in nonpolar covalent bonds and unequal in the polar covalent bond. This depends on the electronegative strength of the participating atoms. Electronegativity is a scale that determines the atoms’ ability to pull the covalent bond electrons.
Atoms with similar electronegative strength (atom’s electronegativity difference < 0.5) share electrons equally. These atoms do not create any opposite poles, and the bond is called a nonpolar covalent bond.
The atoms that form nonpolar covalent bonds are the non-metals and are found on the right side (p-block) of the periodic table. Nonpolar covalent bond examples are - CH4 (electronegativity difference between Carbon and Hydrogen is 0.4), H2, Cl2, I2, etc.
Molecules with nonpolar covalent bonds are very stable and less reactive.
Unequal sharing of the electrons happens when one of the participating atoms in a bond has a higher electronegative strength. Such an atom will pull the bond's electron density more towards themselves, resulting in bond polarity to form a polar covalent bond.
Atoms with high electronegative strength tend to pull the bond electrons more towards themselves to create opposite poles. The atom with more electron density becomes the negative pole, and the atom that loses the electron density becomes the positive pole.
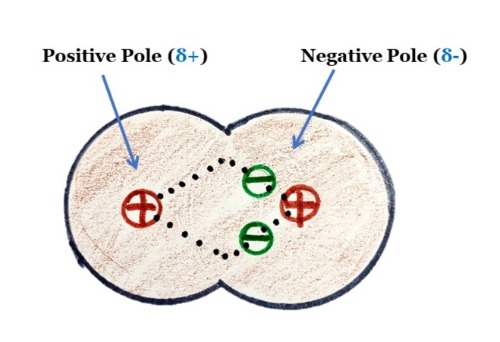
In a polar covalent bond, the atom with higher electron density is shown as delta negative (δ-), and the atom that lost the electron density is shown as delta positive (δ+).
The electronegativity difference between the atoms forming the polar covalent bond is between 0.5-1.7. Some examples of the polar covalent bonds are between the atoms, Carbon and Oxygen, Carbon and Nitrogen, Carbon and Halogen, or between Nitrogen and Oxygen or Halogen.