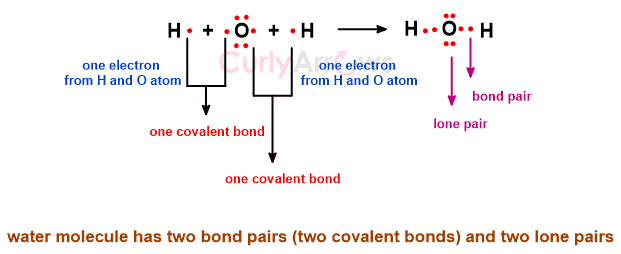
Once the outermost valence electrons of the atoms are engaged in the covalent bond formation to make a molecule, some electrons withhold themselves from the bond-making process. This divides the electrons in the molecule into two parts- bond pair and lone pair.
Bond Pair
The bond pair electrons are involved in the bond formation and are part of the covalent bond holding the atoms.
If only a pair of electrons participated in the bond formation, it forms a single covalent bond. When two pairs of electrons are involved between two atoms, a double covalent bond is formed. Similarly, the participation of three electron pairs from each atom constitutes a triple bond.
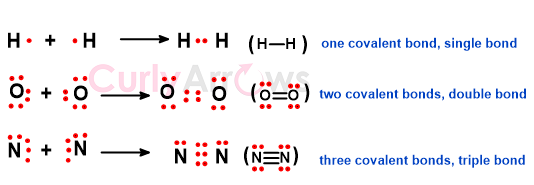
Triple bonds are the strongest, with the shortest distance between the atoms (bond length). It is essentially like tying two ropes with two atoms at each end. Knotting a rope thrice makes the ends (the atoms) come closer and the knot (the bond) stronger.
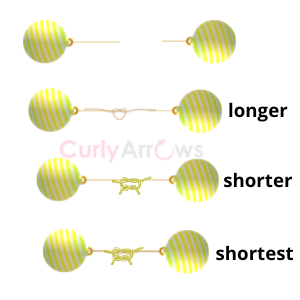
Bond strength: triple> double> single
Bond Length: triple < double< single
Lone Pair
The electrons not participating in covalent bonding remain unshared and are called nonbonding electrons or lone pairs.
The atoms of Oxygen, Nitrogen, and Halogens usually have such lone pairs in their stable compounds. The Nitrogen has one, Oxygen has two, and the Halogen has three lone pairs. The same trend is seen with other atoms of the group they belong to in the periodic table.
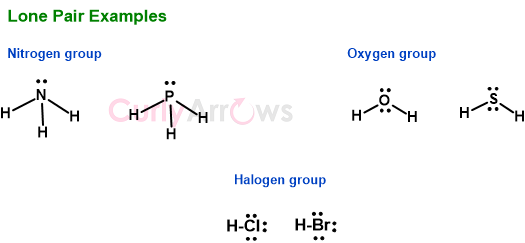
These lone pair are an essential part of an atom and is sometimes involved in forming coordinate (dative) bonds.
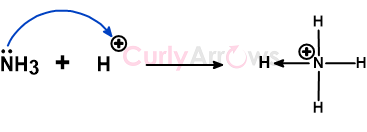
For example, the lone pair on Nitrogen in NH3 can pick up electron-deficient protons (H+), forming a coordinate bond.
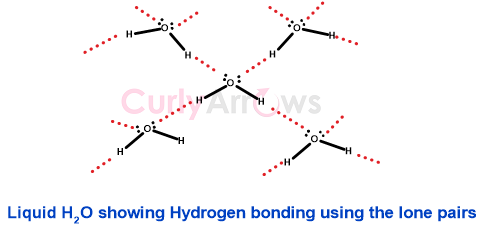
The lone pairs are also involved in an intermolecular attraction that affects the molecules’ physical properties of the melting and the boiling points -for example, Hydrogen bonding.
The water (H2O) molecule is the most common example of hydrogen bonding. The Oxygen atoms’ lone pair has a major role in the liquid nature of water, its high boiling point (100 oC), and the crystalline nature of ice.