Learning Objective: Study Vander Waals London forces, interactions, and strength.
Skill Level – Intermediate
Prerequisites:
Chapter: Intermolecular Forces
Sub-topic: Van Der Waal Forces
Author's Note: Nonpolar molecules have no inherent polar terminals due to the absence of electronegative atoms. Still, they are able to attract due to their constituents – negative electrons and positive protons. Such attraction is universal since all atoms comprise these essential constituents whether or not they have other polar groups. All other interactions are an add-on to the Vander Waals London force. This section covers the most basic yet most crucial attractive interaction occurring universally, where we study how it occurs, its advantages, and factors affecting its strength.
Vander Waals London Dispersion Forces (Nonpolar-Nonpolar Interactions)
Atoms, molecules, or ions interact by attraction or repulsion. Attraction occurs between opposite charges, and repulsion occurs when charges are the same. Therefore, most interactions are controlled by charges.
Understanding interactions is easier when the charges are obvious, like in ions or polar molecules. However, interactions in uncharged, nonpolar molecules occur when the charges originate due to the constant fluctuations of the electrons. The shifting electrons’ position in the outer nuclear region creates polarity, leading to an attractive force, the London dispersion force.

The London dispersion force is the weakest yet most significant Vander Waal forces' subtype, common to all atoms and molecules. The others are Debye and Keesom forces.
Most molecules contain the London dispersion forces in combination with the other prominent forces (dipole, ionic, etc.). So, to study the London Dispersion Forces in isolation- hydrocarbons, homonuclear molecules, or inert gases are used. Such atoms and molecules are essentially nonpolar, therefore, uncontaminated from other interactions arising due to the presence of functional groups or innate dipoles.
A few examples of nonpolar atoms or molecules are- CH4, H2, I2, hydrocarbon molecules (alkanes, alkenes, alkynes), and noble gas atoms (He, Ne, Ar).
Occurrence of London Dispersion Forces in nonpolar molecules
The simplest example used here to understand the London dispersion force is hydrogen (H2), a homonuclear diatomic nonpolar molecule.
H2 is a light gas whose compression into hydrogen cylinders is only possible if it associates by forming close dimers.

In an H2 molecule, there is constant motion and rapid fluctuations in the electron's positions.

Therefore, at any given instant, the molecule can have a situation where most electrons concentrate on one side, creating a temporary negative pole. And the area wherein electron concentration is insufficient gets a positive pole.

The temporary dipole creation in one molecule also urges the nearby molecules of H2 to form temporary poles by creating peer pressure.
They align themselves accordingly and show an attractive induced (forced) intermolecular interaction. So, a few individual molecules’ behavior disperses into a group activity.
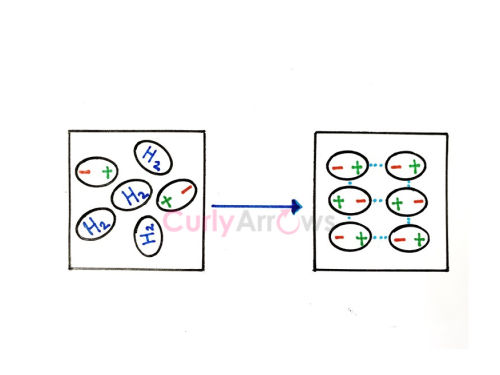
This induced intermolecular attractive interaction type is known as the London Dispersive Force, named after physicist Fritz London. It is also known as an induced dipole-induced dipole type of interaction.
Importance of London Dispersion Force in liquefaction of gas
The attractive interaction due to the London Dispersion Forces brings otherwise isolated atoms, and molecules close so that they change state from gas to liquid to solid on the application of lower temperature and high pressure, a process known as liquefaction.
The conversions are usually done under a vacuum condition that discourages any interference from the air or other solvents. The application of vacuum highly favours close atomic contact and liquefaction after that.
Under such conditions, the molecule's kinetic energy decreases, reducing its mobility. For example, in the case of liquefaction of H2, the gas must be cooled to a very low temperature of -252.87 oC, which slows down its kinetic energy and converts it to a liquid state.
Similarly, neutral atoms like He, Ne, Ar, and butane can also be compressed and liquefied to be sold as Helium/Argon/ Nitrogen or as LPG gas cylinders for home and commercial purposes.
London Dispersion Forces– Role of atomic size
Due to the intermolecular London dispersive type of attraction, polyatomic nonpolar molecules like CH4, SiH4, and GeH4 can condense to the liquid and the solid-state under low temperature and high-pressure conditions.
However, the strength of these interactions is dependent on the atomic size.
The larger, heavier atoms of polyatomic molecules have more electron orbitals, contributing to their size. Here, the electrons are dispersed over a larger surface area and can be easily influenced to arrange themselves into opposite poles, thereby showing significant interactions. However, the nuclear charge firmly holds fewer outer electrons in smaller atoms, decreasing its ability to form poles.
As seen from their boiling point values, the greater attraction in the molecules with bigger atoms translates to these compact clusters of molecules requiring more energy to break their interactions, raising their melting and boiling point.
For example,
Vander Waal’s strength - CH4< SiH4< GeH4 since, size of C<Si<Ge.
Boiling Point- 112K <161K <183K

Similar effect of the attraction on size is seen in the halogen series. Comparing the halogen series (F2, Cl2, Br2, I2) with their sizes, it is observed that the nonpolar, larger I2 interacts strongly and therefore is solid compared to F2 or Cl2.
I2 atoms, due to their large size and more electrons, can cause significant distortion in their electron cloud, creating greater intermolecular attractive interactions compared to the small atoms of F2 or Cl2.

Therefore, the melting point of I2 is significantly higher than F2 and Cl2.
Van der Waals Strength: I2>Br2>Cl2>F2
Melting point: 113.7oC> -7.2oC> -101.5 oC> -219.67 oC
London Dispersion Forces– Role of atomic shape
In nonpolar hydrocarbon molecules of alkanes, the shape of the molecules determines the strength of the interactions.
A long linear hydrocarbon molecule can disperse electrons over a larger region than compact spherical nonpolar hydrocarbons.
The scattered electrons are like hands grasping the nearby molecules and packing tightly, allowing for stronger attractive molecular interactions. These interactions also raise the melting and the boiling point.

For example, comparing the linear n-pentane with spherical neo-pentane, with nearly the same number of Carbon-Hydrogen atoms (C:H ratio, 5C:12H), the boiling point of n-pentane is 36.2 o C, and that of neo-pentane is 9.5 o C.
Therefore, the order of Vander Waal’s strengths is -
CH3-C(CH3)2-CH3 < CH3-CH2-CH2-CH2-CH3
Subscribe to study all types of Intermolecular Forces.
