The Covalent bond is one of the two linkages by which atoms join to form molecules and is therefore classified under chemical bonds.
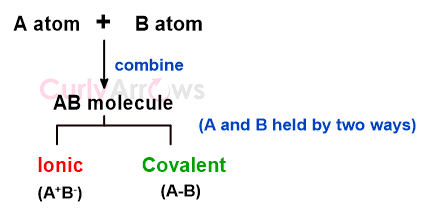
Not all atoms can choose to form covalent bonds and are only seen between two nonmetal atoms. Being in proximity also doesn’t guarantee bond formation, since atoms regularly bump into each other, and not all collisions have led to a molecule formation.

The chance of bond formation is high when the approaching atoms have a correct orientation, high concentration, and sufficient velocity for colliding.
The orientation, concentration, and speed can help in two ways- most atoms will now possess the minimum energy (called the threshold energy) required to overcome the interatomic electron repulsions and come closer. And once closer, they will be attracted due to their opposite electrostatic nature- the negative electron of one atom with the positive nuclei of the other.
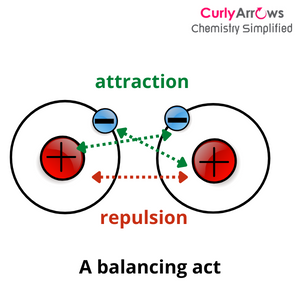
Once these parameters are in order, the atoms then reach a favorable distance, called the bond length. At this length, atoms’ attractive and repulsive forces are balanced and are ready for an embrace.
.png)
Each atom shares one electron, overlapping to form a two-electron covalent bond.
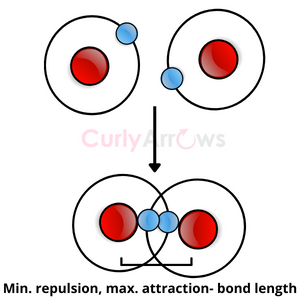
The two electrons are now part of both atoms’ nuclei and keep moving between them.

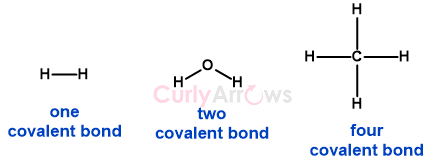
The joining of atoms by a covalent bond is shown as a dash (-). For example, H-H is a covalent bond between two atoms of Hydrogen. H-O-H is a covalent bond between Oxygen and Hydrogen, where Oxygen has shared two electrons to form two covalent bonds with two Hydrogen. CH4 is Carbon, sharing its four electrons with four hydrogen atoms to form four covalent bonds.
The energy the atoms acquire for the bonding process; the excess energy is released as heat once the covalent bond formation completes. Therefore, covalent bond formation reactions are exothermic (heat-producing) in nature.
Apart from collisions, the other ways the atoms approach and form covalent bonds are by - electrostatic attractions or reactions driven by harsh conditions of temperature, pressure, radioactivity, or short-wavelength light (for example, microwave reactions, radical reactions in UV light).
Covalent bonding holds less stable, free atoms in stable, strong, fixed bonds.