Sharing resources is essential to build a harmonious world. When the resources are shared ineffectively conflicts emerges. A similar principle extends to Chemistry.
Atoms are in a state of harmony when the neighbouring atoms shares electrons. When the sharing stops, Chemical reactions triggers.
The imbalance in sharing electrons influences the molecule’s polarity, reactivity, and physical properties. The disagreement on sharing is brought about by electronegativity.
Introduction to electronegativity
A covalent bond is made of two electrons. When similar atoms form a molecule, they harmoniously share the bond electrons (e.g., H2, Cl2, N2).
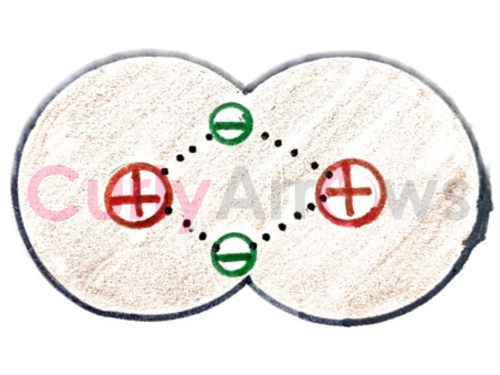
But when different atoms like H-Cl, H-O, and N-H join to form molecules - H-Cl, H2O, NH3, the electrons in the bond are shared unequally.
The unequal sharing is when one atom pulls the two bond electrons more strongly towards itself, resulting in the atom getting more of the electron density.
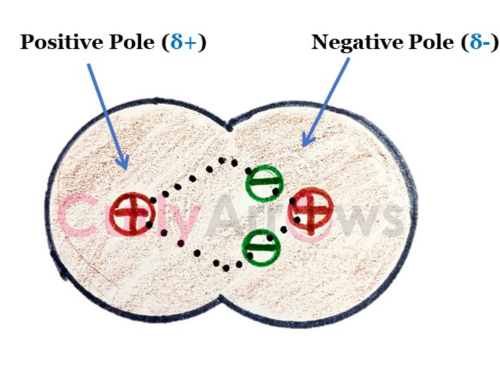
This ability of an atom to pull the bond electrons is called electronegativity. The atom that pulls the electrons more strongly is denoted as delta minus (δ-) and is more electronegative. Whereas the other atom, due to the loss in electron density, gets delta plus charge (δ+) and is said to be less electronegative.
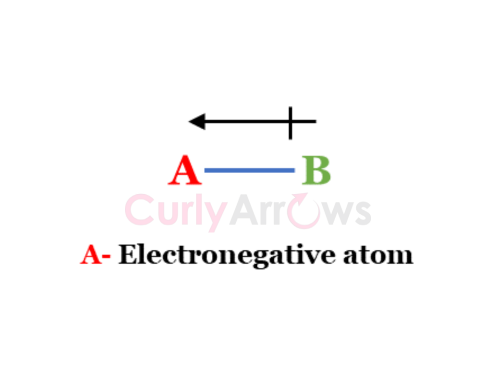
Sometimes an arrow is drawn over the two atoms to indicate the directional nature and the magnitude of the electron pull. The arrowhead rests on the electronegative atom and the crossed tail on the less electronegative atom.
Pauling devised an electronegativity scale, denoted as (Χ, chi - pronounced as Kai) based on an atom’s ability to pull the bond electrons, known as the Pauling scale. The scale ranges from 0.79 to 3.98 and is a unitless quantity.
Lower values are for the least electronegative atom (Ex: metals), and a higher value is for the most electronegative atom (Ex: the halogens). Amongst the metals, Caesium (Cs) has the lowest electronegativity, and Fluorine (F) has the highest of all the halogens.
Pauling arrived at these arbitrary values by calculating the energy required to break the two-atom bonds.
A-B
Where A, B- are the elements from the periodic table.
From the bond dissociation energy values, he assigned Fluorine the highest electronegativity value of 4, and the other values were calculated relative to it.
Electronegativity values of common elements from the periodic table
Electronegativity values of some familiar elements from the periodic table are as:
| Hydrogen (2.1) | Carbon (2.5) | Nitrogen (3.0) | Oxygen (3.5) | Fluorine (4.0) |
|---|---|---|---|---|
| Lithium (1.0) | Phosphorus (2.1) | Sulfur (2.5) | Chlorine (3.0) | |
| Arsenic (2.0) | Selenium (2.4) | Bromine (2.8) |
The most important and commonly encountered ones in the decreasing order of their Electronegativity are listed along with a fun learning mnemonic:
F> O> Cl> N> Br> I> S> C> H> metals
FOCused NeighBour Ian iS Collecting Historical Metals
The electronegativity difference between the bonded atoms only causes partial charge separation, unlike ionic bonds wherein the electron transfers are complete. The electronegative atoms are shown as δ+/- charges whereas total charges (+/-) are shown for the positive or negative ions. E.g. Na+ and Cl- are ions, but CH3(δ+)-Br(δ-) represents partial charge separation.
Therefore, the electronegativity concept applies to all the atoms in a covalent bond state and does not apply to free atoms.
This is an excerpt from CurlyArrows' Introduction to Organic Chemistry Premium Tutorials, Chapter Electronic Displacements in a Covalent Bond.
