
Pre-Requisite Reading: Resonance
Resonance is an integral concept that is often used to explain the extra stability that certain molecules possess, attributed to electron delocalization. It is observed that the molecules that delocalize electrons are more stable, and such molecules also tend to have more than one Lewis structure.
This stability is measured in resonance energy or delocalization energy.
How is resonance energy calculated?
As an example, let’s consider a molecule like Benzene, a classic example of resonance. To understand why Benzene is an epitome of resonance, we must theoretically and experimentally break it down into gaseous atoms to observe the difference in the released energy. The difference in energy is attributed to the remarkable electron delocalization or resonance stability in the molecule.
Benzene is a six-carbon cyclic molecule with alternating single and double bonds. Theoretically, it is a cyclohexatriene, a cyclic hexagon ring with three alkene bonds.
Cyclohexatriene has three components-
3 Carbon-Carbon double bonds, 3 Carbon-Carbon single bonds, and 6 Carbon-Hydrogen single bonds.
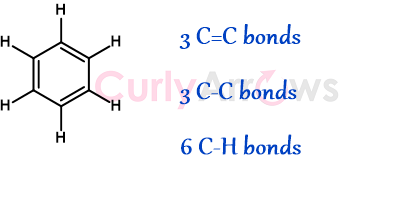
It is assumed that when a cyclohexatriene is heated, and its bonds are broken to liberate gaseous atoms of Carbon and Hydrogen, 5390 kJ/mol of energy is released.
The theoretical value of 5390 kJ/mol is obtained using the data from the previous standardization experiments. The standardization experiments calculated the heat of atomization values of some carbon bond types - C-C single bond, C=C double bond, C-H single bond, etc. by breaking each bond type into gaseous atoms. The energy released indicates the bond strength and these values were then used for the cyclohexatriene molecule.

However, breaking down Benzene into gaseous atoms (heat of atomization) of Carbon and Hydrogen is found to have a value of 5535 kJ/mol. Subtracting the value of cyclohexatriene 5390 kJ/mol with 5535 kJ/mol for Benzene gives 145 kJ/mol. This extra 145 kJ/mol of energy is the resonance energy and is obtained due to electron delocalization or resonance.
Therefore, resonance energy is the prime factor differentiating isolated bonds of cyclohexatriene from Benzene by imparting additional stability.
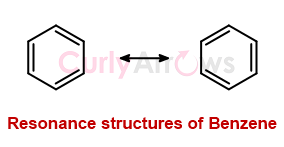
How is resonance energy calculated in other molecules?
In most cases, the resonance energies are calculated indirectly or are estimations.
If a molecule has multiple resonance structures, the structure with the lowest energy is used for calculation. Such a structure is arrived at by following the Resonance structure drawing rules. These rules, known as the rules of resonance, predict a molecule's most stable, lowest-energy Lewis structure.
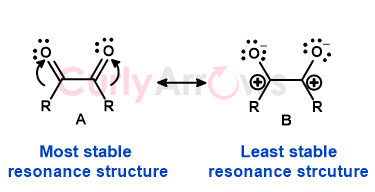
The resonance energy is then the difference in energy between the actual molecule and the most stable hypothetical Lewis structure.
Note that, in reality, the resonance energy of the actual molecule is always greater than any of the predicted Lewis structures.
Higher resonance energy always correlates with higher electron delocalization and larger stability.
The resonance energy of some other molecules is Naphthalene (255 kJ/mol), Pyridine (117 kJ/mol).
Take the Quiz
