Arguments in favour of NF3
The Dipole moment is a product of charge difference (q) and the distance (d) between the centres of positive and negative charges (µ = q x d, SI unit Debye/Coulomb meter); the following observations support similar or slightly higher dipole moment for NF3:
a) the electronegativity difference between N-H and N-F is similar (0.9 for N-H and 1.0 for N-F),
b) Fluorine and Hydrogen have comparable atomic sizes (Fluorine 42 pm and H 53 pm), with the bond distance for N-H and the N-F being 100 pm and 137 pm.
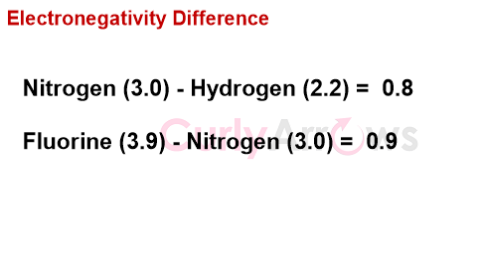
These parameters lead to the expectation that the dipole moment of NF3 should be like NH3 or slightly higher than NH3; however, the observed value is 1.46 Debye for NH3 compared to 0.24 Debye for NF3.
Role of Molecular Geometry
The deviation is due to the Dipole moment calculation not including the molecular shape (molecular geometry/symmetry), which is crucial in determining the net dipole moment.
The molecular Dipole moment is a vector quantity that considers direction and magnitude. Both molecules have a trigonal pyramidal shape; however, their bond moments' direction differs according to the atoms' electronegativity since the net dipole moment is always the sum of all the individual bond moments. For complex molecules like NH3 and NF3, the net dipole moment also considers the contribution of the lone pair.
NH3 molecule
The direction of the vector for the N-H bond moment lies towards the electronegative Nitrogen. The positive end is Hydrogen. The orbital lone pair is more electronegative than the Nitrogen and pulls the electron cloud towards itself. The vector, therefore, points in the direction of the lone pair. The resultant N-H bond moments reinforce the moment of the lone pairs leading to a more significant net dipole moment.
NF3 molecule
The direction of the bond moment for the N-F bond is more toward the electronegative Fluorine. The positive end of the vector faces the Nitrogen. The vector points toward the lone pair as it is more electronegative than the Nitrogen. The resultant N-F bond moments are opposite to the bond moment of the lone pairs. Therefore, the bond moments that are oppositely faced cancel each other out, and the net dipole moment for NF3 is less.