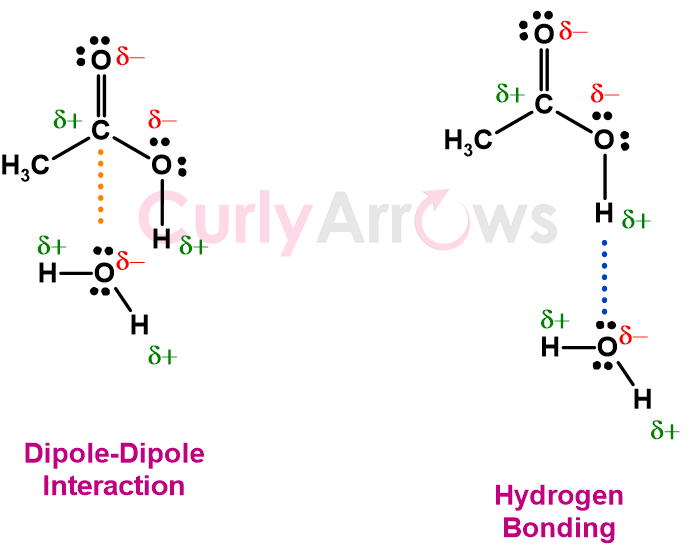
Dipole-dipole interactions occur in polar molecules where the difference in electronegativity between the combining atoms creates positive and negative dipoles. These opposite poles align and result in electrostatic attraction throughout the polar medium. So, naturally, the strength of the interaction would depend on Coulomb’s law - the higher the magnitude of the charges and the lesser the distance between those charges, the stronger the dipole-dipole attractive interaction.
Hydrogen bond, however, depends less on the Coulombic law. Instead, there is greater reliance on the bond angle of the Hydrogen bond and the covalent bond linking the donor hydrogen to the electronegative atom of the donor.
Hydrogen bonds are at their strongest when the bond angle is 180°. The strength of the bond decreases rapidly as the bond angle deviates from this ideal angle. Such a dependence on the bond angle is because the Hydrogen bond formation is partially covalent in nature, involving slight electron sharing between the Hydrogen donor and the lone pair of the electronegative acceptor atom. Such electron sharing enables the orbitals of the atoms to overlap, encouraging stronger bonding, which then comes with bond angle restrictions.
The partially covalent character of hydrogen bonds is the main reason why these interactions are stronger than other dipole-dipole interactions.
The strength of hydrogen bonds typically ranges from 0.2 to 10 kcal/mol, whereas other dipole-dipole interactions are roughly a tenth as strong, ranging from 0.02 to 1 kcal/mol.
Although hydrogen bonds are weaker than covalent bonds, they have the advantageous ability to form networks. The crystalline structure of ice is a result of hydrogen bonding, which forms empty pockets that allow ice to float on water. Water is a universal solvent due to hydrogen bonding, and this phenomenon even extends to biology by affecting the shape of DNA and the binding of proteins, thus influencing the molecules of life.
