
Understanding the structure of NO2
The Nitrogen has five valence electrons in its outermost shell (shown enclosed in the blue circle). The two out of the five electrons donate both electrons to Oxygen and form a dative (coordinate) bond. The other two electrons (on the right) of the Nitrogen are shared with the Oxygen to form a double bond. The remaining electron is the free radical, and NO2 molecules’ paramagnetic behavior proves its presence. The magnetic field weakly attracts the NO2 molecules due to its unpaired electron.
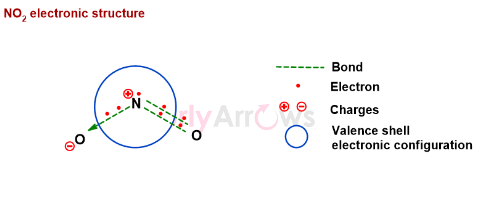
The free radical electron is electron-deficient specie. They are unstable and high in energy. The NO2 molecule tries to lower the high energy by forming a bond with itself so that the unpaired electron of the free radical is paired.
A bond where two molecules of the same species combine to form an adduct with a new covalent bond, such a molecule is called a dimer.
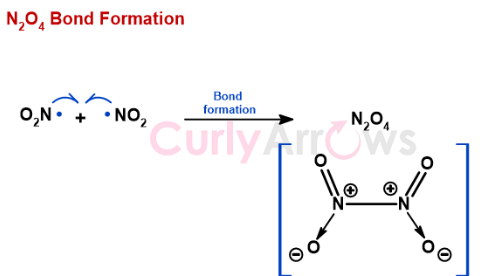
Once the free radical pairing is complete, the molecule N2O4 has no high- energy electrons left. N2O4 becomes diamagnetic and is repelled by the magnetic field.
Importance of Dimerization
The electron pairing allows the Nitrogen to attain a stable octet configuration of the nearest inert gas configuration of Neon (two core electrons and eight valence electrons) and brings stability to the molecule.
The Lewis structure of N2O4 shows the complete stable electronic configuration.
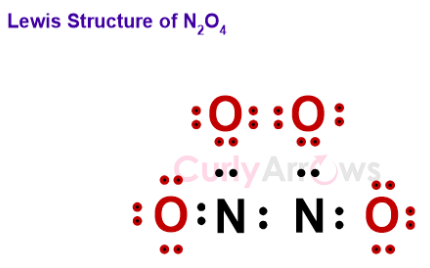
Dimerization reaction that lowers the energy and brings stability is a highly favorable process.
This excerpt is additional content inspired by the chapters Reactive Intermediates, Bonding in Atoms, and Drawing Organic Structures, which are part of CurlyArrows' Introductory Organic Chemistry Course. Preview the Book.
Related Reading-
Free Radical, Introduction, Structure, and Types
Difference between a Free radical and an Electrophile
