Benzoic acid does not undergo specific Friedel Craft Reactions. The aim is to subject benzoic acid to Friedel Craft Acylation and Friedel Craft Alkylation Reactions by using it in two ways - as a substrate and as a reagent to help find favorable reaction conditions.
Friedel-Craft Acylation Reaction
It is an electrophilic aromatic substitution reaction used to introduce an acyl group (R-C=O) in the aromatic ring resulting in aryl ketones.
The reacting molecules are benzene (substituted or unsubstituted), an acyl halide (any RCOCl as the source of the acyl group), and a Lewis Acid catalyst (for example, AlCl3, SnCl4, TiCl4, BF3, FeCl3, etc.).
General Reaction
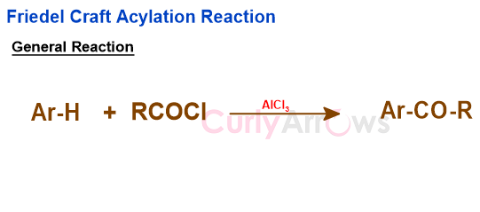
Benzoic acid as the Substrate for Friedel Craft Acylation Reaction
Benzoic acid is an electron-withdrawing group that deactivates the Benzene ring by pulling away from its electron density. Friedel-Craft Acylation Reaction prefers electron-donating or ring activating groups in the substrate. Therefore, benzoic acid or any other electron-withdrawing groups are not a preferred substrate for the Friedel Craft Acylation reaction.
Only electron-donating groups or substitutions that increase the Benzene ring electron density by either +I (positive inductive effect) or + R (positive resonance effect) favours Friedel-Craft Acylation Reaction. For example, alkyl, alkoxy, halogen, etc.

Benzoic acid as the Reagent (Source of the Electrophile) for Friedel Craft Acylation Reaction
Benzoic acid in strong acids generates acylium ion (Ph-C≡O +) that is stable resonance. It is a good source of electrophile for the Friedel-Craft Acylation reaction. The acylium ion of the Benzoic acid successfully generates aryl ketones (Ar-CO-R).
The acids that help undergo this transformation are -PPA (polyphosphoric acid), H2SO4 (sulfuric acid), TFA (trifluoroacetic acid).

The more reactive forms of carboxylic acid - acid chloride (Ph-CO-Cl) or acid anhydride (Ph-CO-O-CO-Ph) can also be used as a source of acylium ions.
However, suppose the carboxylic group (-COOH) is not directly attached to the phenyl ring but is a part of the alkyl functional group. In that case, it can then undergo an intramolecular cyclization reaction by the FC acylation method. Intramolecular cyclization would depend on the length of the alkyl chain.

Friedel-Craft Alkylation Reaction
It is an electrophilic aromatic substitution reaction that introduces an alkyl group (-R) in the Benzene ring to form alkylbenzenes.
The molecules taking part in the reaction are -benzene (substituted or unsubstituted), an alkyl halide (RX as the source of the alkyl group), and a Lewis Acid catalyst.
General Reaction:

Benzoic acid as the Substrate for Friedel-Craft Alkylation Reaction
The carboxylic group (-COOH gr) functional group as a part of the Benzene ring is electron-withdrawing and, therefore, deactivates the ring for any electrophilic substitution reactions. Therefore, the Benzoic acid does not undergo Friedel-Craft Alkylation Reaction.
