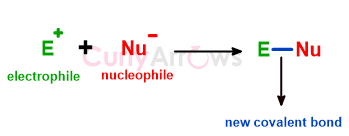
Electrophiles are electron-deficient species that accept electrons from other electron-rich counterparts, the nucleophiles, to form a two-electron covalent bond.
The sign +/δ+ denotes the electron deficiency. A positive charge (+) implies a complete lack of two electrons. At the same time, the delta positive (δ+) sign indicates a polarity difference created by the adjacent electronegative atom that pulls the bond electron density towards itself, creating an electron-deficient δ+ center.

There are also uncharged electrophiles that can accept electrons due to their vacant or extra d-orbitals. Examples are- BF3, PCl3, etc. Or atoms with incomplete octet electron configuration like in carbene, nitrenes, and free radicals.
Read more about the Electrophiles.
