The mass number is the total count of the number of protons and neutrons present in the nucleus of an atom, denoted as A.
For example, 12C has 6 protons and 6 neutrons; its mass number is 12.
A mass number is always a whole number. The mass number is also known as the atomic mass number or nucleon number.
The isotope of an element has the same number of protons but a different number of neutrons. Therefore, the mass number of each isotope of an element will be different.
For example, Carbon has three isotopes- 12C, 13C, and 14C. 12C has a mass number of 12, a sum of its 6 protons and 6 neutrons. 13C has 6 protons and 7 neutrons; its mass number is 13. 14C has 6 protons and 8 neutrons; its mass number is 14.
The mass number is written in the superscript of the AZE notation while representing an element.
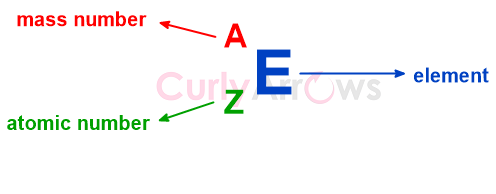
The mass number is different from the standard atomic weight of an element that takes into account the average of all the isotopes of an element. In comparison, the mass number counts the protons and the neutrons for each isotope separately.
20 elements from the periodic table have no naturally occurring isotope and exist in only one form.
For example, all Fluorine atoms found in nature have 9 protons and 10 neutrons. Their mass number is 19.

Other naturally occurring elements have more than one isotope. For example, the element tin (Sn) has 50 protons, but tin atoms can have 62, 64, 65, 66, 67, 68, 69, 70, 72, or 74 neutrons. Therefore, tin has 10 natural isotopes with mass numbers of 112, 114, 115, 116, 117, 118, 119, 120, 122, and 124.
