Nucleophiles are an atom or a group of atoms that are richer by two electrons and donate these electrons to electron-deficient species, the electrophiles.
Donating the electrons from the nucleophile to the electrophile creates a new two-electron covalent bond.
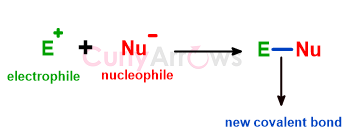
The electron richness of the nucleophile is evident in atoms carrying extra electrons in the form of a negative charge, lone pairs, pie bond, or sigma bond.

The atoms that are good nucleophiles are usually heteroatoms of Nitrogen (N), Oxygen (O), Sulfur (S), or Halogen (X). Example, NH3, H2O, OH-, X-, HS-, etc.
The other less stable but strong nucleophiles are Hydrogen (e.g., H-) and Carbon (e.g., CN-, RMgX, RLi).
Read more about the Nucleophiles.
