Keesom Forces | Debye Forces | London Forces |
|---|---|---|
The Keesom interactions occur in polar molecules with permanent dipoles. | Debye type of interactions occurs between the polar and nonpolar molecules. | Nonpolar molecules only interact by the London Forces.
|
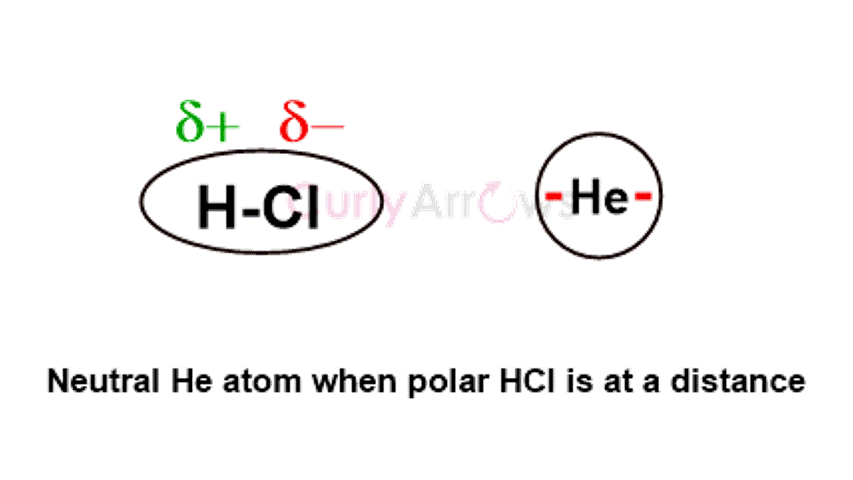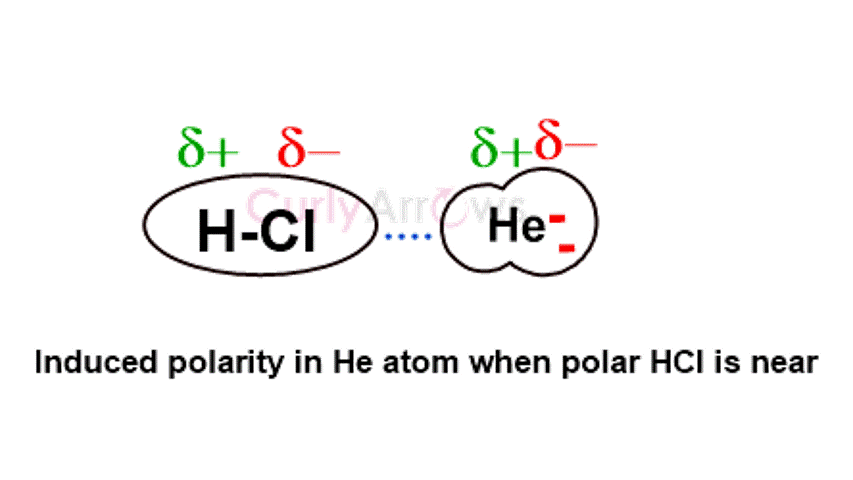
The electrostatic interactions align the permanent dipoles of the polar molecules, causing an orientation effect. | The dipoles of the polar molecule induce the formation of polar terminals in the nonpolar molecules bringing about an induction effect. | The rapidly changing positions of the electrons cause an asymmetrical distribution, forming instantaneous dipoles in a few nonpolar molecules. This forces temporary dipole creation in nearby molecules, producing a dispersion effect. |
It is also known as Dipole-Dipole interactions. | It is also known as Dipole-Induced dipole interactions. | It is also known as Instantaneous Dipole- Induced Dipole interactions. |
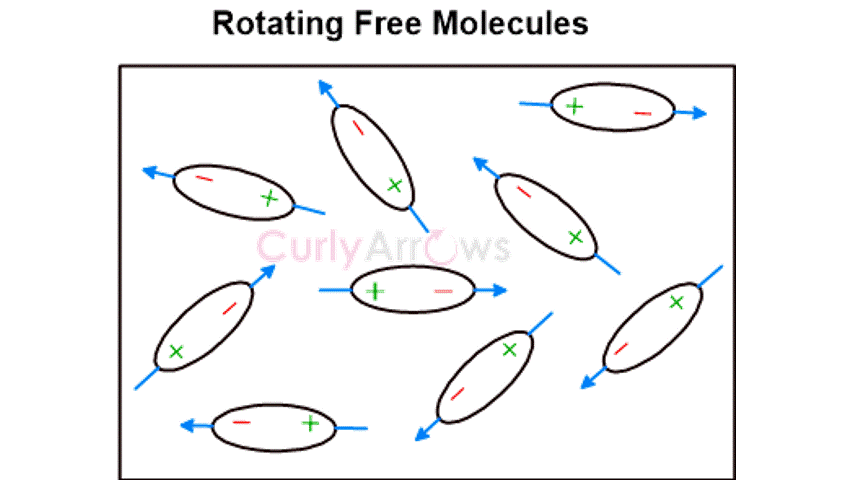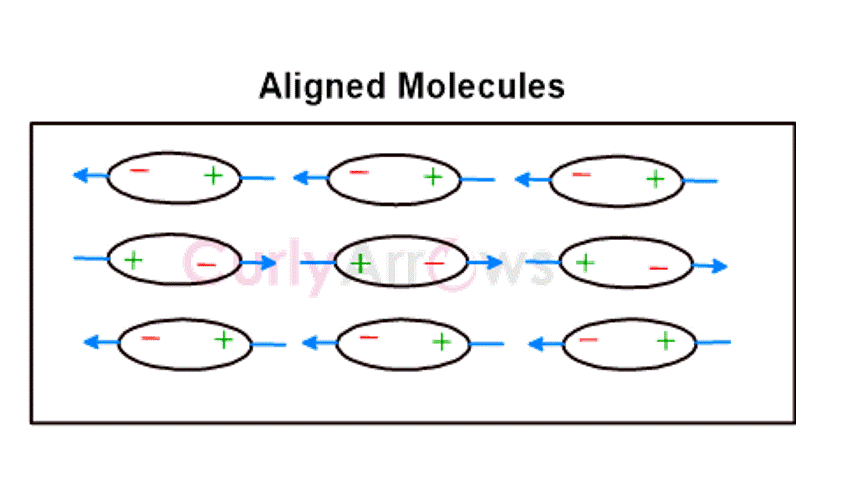 |  | 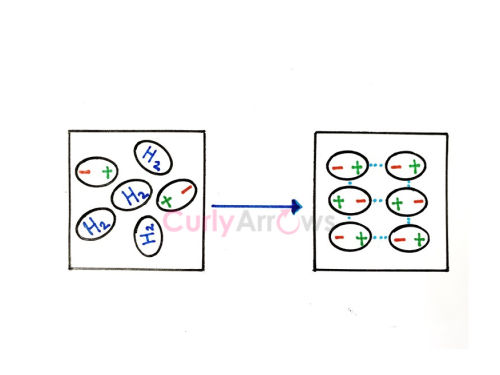 |
Keesom forces are the strongest among all intermolecular van der Waal forces of interactions. | Debye forces are stronger than London forces but weaker than Keesom forces of intermolecular interactions. | London forces are the weakest among all the three types of van der Waal forces of interactions. |
Polar molecules with heteroatoms like H2O, HCl, etc., show the Keesom effect. | A combination of a polar molecule like HF, H2O, and a nonpolar atom like Ar, He, N2, etc., shows this effect. | Nonpolar molecules like H2, CH4, Ne, N2, etc., show this effect. |
Related Reading: Keesom Force, Debye Force, London Dispersion Force.
