Why Anti-Markovnikov's Rule is important for alkenes?
Alkenes undergo addition reactions where it loses one pie bond to form two new sigma bonds to add a reagent across the double bond.
Symmetrical alkenes undergo an addition reaction and form one dominant product. However, confusion arises when it is an unsymmetrical alkene. The reagent can be added on either end of the double bond to form more than one product.
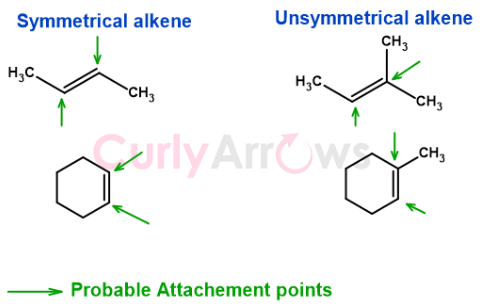
Markovnikov solved the regiochemistry dilemma in 1870 by proposing the Markovnikov rule.
Few reagents such as Hydrogen Halides (HX), Sulfuric Acid (H2SO4), Alcohols (R-OH), and Water (H2O) follow Markovnikov's Rule for the addition across the double bond of an unsymmetrical alkene.
However, HBr in the presence of a peroxide showed a behavior opposite to Markovnikov's rule of alkene addition, thereby termed ‘The Anti-Markovnikov's Rule.’
In Anti-Markovnikov addition, the negative part of the reagent attaches itself to the least substituted Carbon of the double bond. The least substituted Carbon is the Carbon with most Hydrogen atoms.
The reaction was studied extensively by Morris Kharash and was explained in the paper titled: ‘The Addition of Hydrogen Bromide to Allyl Bromide' in 1933. As a tribute to his work, the Anti-Markovnikov rule is also known as the Kharasch effect. The reaction works only in the presence of peroxide; therefore, it is also called the ‘Peroxide effect.’
The Anti-Markovnikov addition to the double bond is shown only by HBr and not HF, HCl, or HI, even in the presence of the peroxide.
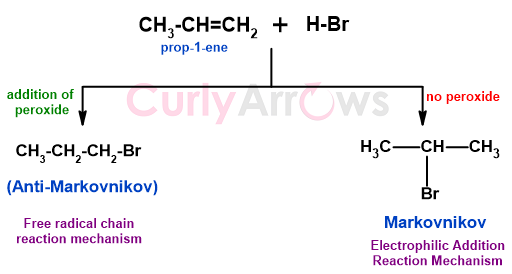
HBr addition follows old Markovnikov's Rule if the peroxide is absent.
Why does HBr add differently in the presence and absence of the peroxide?
There is a difference in the reaction route taken by HBr in the presence of peroxide and its absence. In its absence, HBr adds to the double bond by electrophilic addition mechanism (using Markonikov's Rule). However, in the presence of peroxide, the addition of the HBr occurs via a free radical chain reaction.
Anti-Markovnikov Free-Radical Reaction Mechanism
The free radical reaction mechanism occurs in three steps - Initiation, Propagation, and Termination.
A) Initiation
The initiation step requires any one of the radical generators - Hydrogen peroxide (H2O2), Organic Peroxides (R-O-O-R), Benzoyl peroxide (C6H5-CO-O-O-CO-C6H5), Organic Hydroperoxides (R-O-O-H), Azobisisobutyronitrile (AIBN), etc. of which most employed are the peroxides.
In the first step, the alkyl peroxides (R-O-O-R), when exposed to UV light or heat, undergo homolytic cleavage to generate two alkyloxy radicals (2R-O.).
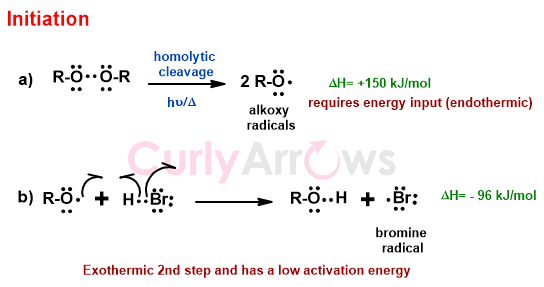
The alkoxy radical then picks up the Hydrogen from H-Br to form a Bromine radical (Br.) that reacts with the unsymmetrical alkene (for example, 1-propene) in the next step.
B) Propagation
The Bromine radical can attach itself to either side of the double bond. If it attaches to site (i), the alkene forms a 10 radical. If Br. Attaches to the site (ii), in that case, a 20 radical of the alkene is obtained.
The 2o radical is more stable than the 1o radical. Since the radical is short of one electron, it is an electron-deficient specie. Therefore, it follows the stability order like the carbocations-
30> 2o> 1o> CH3
Thus, the Bromine radical chooses to attach at the site (ii) to form a stable 20 alkene radical over the less stable 10 alkene radical.
In the next step, the stable 2o radical picks up the H from the HBr to form a bromoalkane (1-bromopropane). This step also generates a new Bromine radical to propagate the chain reaction. These two steps continue until the reaction reaches the termination stage.
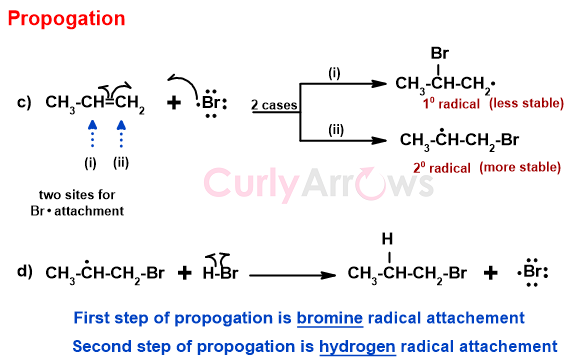
(Note that in the Anti-Markovnikov method, the addition of Hydrogen is the second step in contrast to the first step in Markovnikov's Rule.)
C) Termination
The reaction discontinues when the free radical species (bromo and alkyl) are exhausted by one following steps-
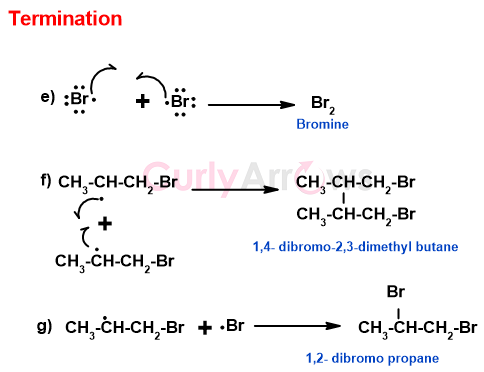
The radical termination reactions give an insight into the nature of the products and the by-products formed. The by-products are separated from the main products by employing various purification methods.
Therefore, Anti Markovnikov's addition of HBr on an alkene gives a bromoalkane with the halogen attached to the least substituted carbon.
An important point to remember is that, in both the Markovnikov and Anti-Markovnikov's Rule, the stability of the intermediate carbocation and the radical dictates the regio-chemistry of the product; that is, the decision on where the halogen must attach.
Other reactions following Anti-Markovnikov’s Rule of Addition
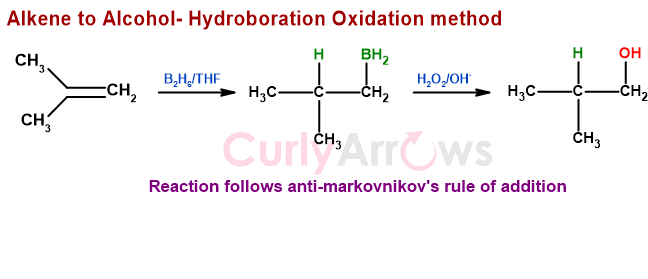
The addition of water across the double bond of an alkene follows the Anti Markovnikov's Rule when the reagents are Diborane (B2H6) and peroxide in solvent THF. The reaction generates alcohol at the less substituted Carbon. The first step of the reaction is hydroboration, followed by the oxidation step.