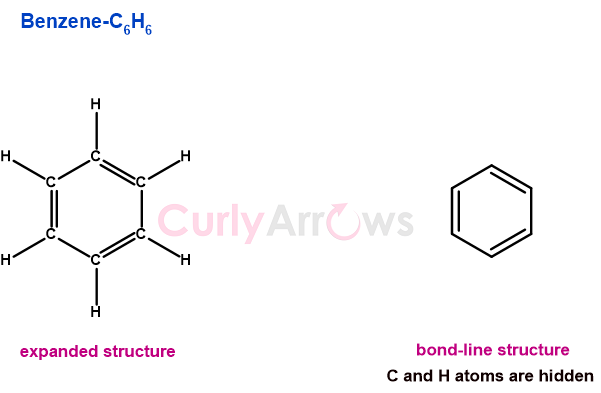
Bond Order for Covalent Bonds
The very premise of a covalent bond is electron sharing. As two atoms share one electron each to form one covalent bond, they may likely share more than once and form more bonds. This information on the number of connections between two atoms is revealed from the Bond Order.
So, the Bond Order measures the number of bonds between the two atoms in a molecule. The number can be integers like 1, 2, or 3 for single, double, or triple bonds or non-integers like 0.5, 1.3, 1.5, etc.