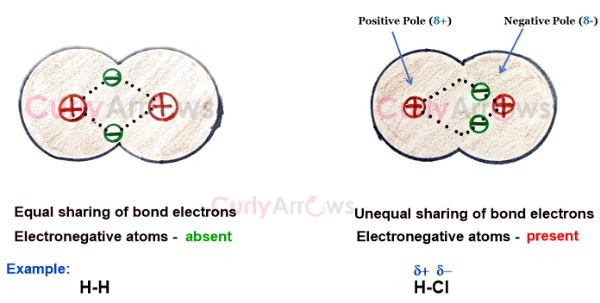
Electronegativity measures on a scale of 0.8 – 4 an atom’s or group of atoms’ tendency to attract the bond electron pair towards itself, thereby creating partial negative (δ-) and positive (δ+) terminals.
The electronegativity value is a unitless quantity denoted as Χ (chi, pronounced as kai).
Sometimes a dipole arrow is used over the bond to indicate the direction and magnitude of the electron movement. The arrowhead rests on the more electronegative atom, and the arrow's positive end on the less electronegative atom.

Electronegativity difference is used to classify the chemical bonds into- nonpolar covalent, polar covalent, and ionic bonds.
Read more about electronegativity.
