Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields
i) A single monochloride
ii) Three isomeric monochlorides
iii) Four isomeric monochlorides.
Prerequisite Reading: Structural Isomerism, Types of Hydrogen, Free radical halogenation
The compound C5H12 is a saturated alkane following the general formula of CnH2n+2, where n is the number of Carbon atoms. The different chain isomers that are possible for the given molecular formula are-
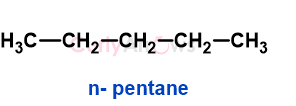
a) n-pentane
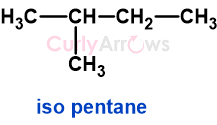
b) Isopentane
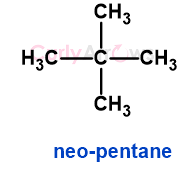
c) Neo-pentane
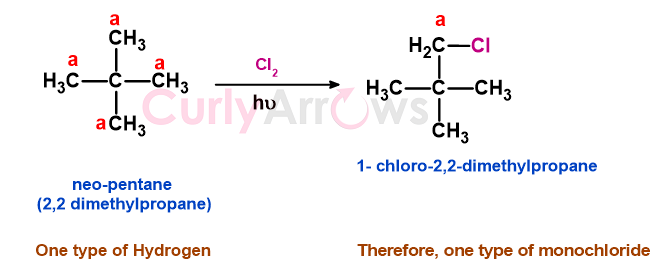
Depending on the number of types of Hydrogen present, the chain isomers on photochemical chlorination will yield single or multiple monochlorinated positional isomers -
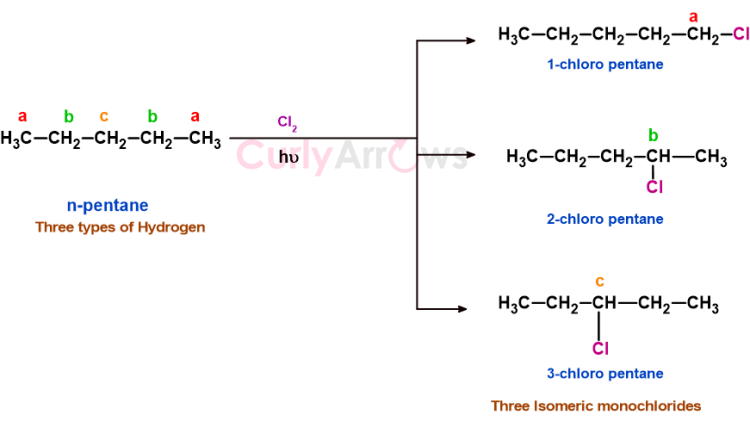
a) n-pentane has three types of hydrogen that on monochlorination gives 1-chloropentane, 2-chloropentane, and 3-chloropentane.
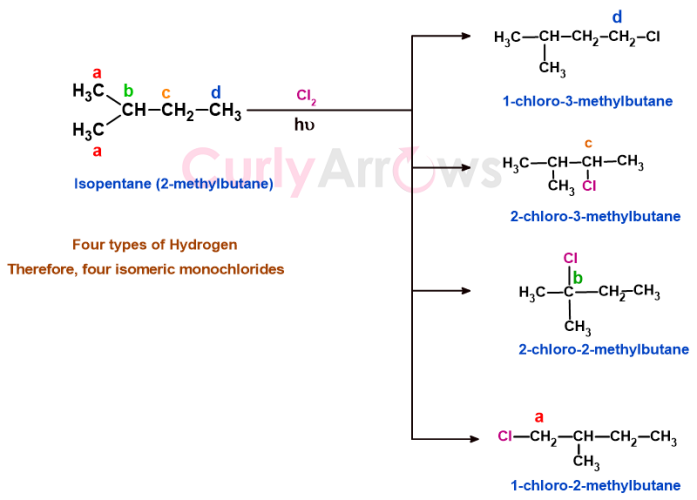
b) Isopentane has four types of hydrogen that on monochlorination gives 1-chloro-3-methylbutane, 2-chloro-3-methylbutane, 2-chloro-2-methylbutane, 1-chloro-2-methylbutane.
c) Neopentane has one type of hydrogen that on monochlorination gives 1-chloro-2,2-dimethylbutane.