Friedel Craft Acylation reaction is an electrophilic aromatic substitution reaction involving acylium ion as the electrophile.
A new Carbon-Carbon bond is formed in the reaction by replacing the one hydrogen with the acyl group, thereby introducing a ketone functionality in the molecule.
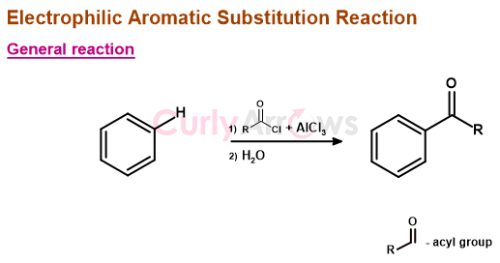
General Reaction – FC acylation
The FC acylation reaction requires three reagents: Benzene (as the nucleophile), acyl chloride, and Lewis acid AlCl3 (to generate the electrophile, acylium ion).
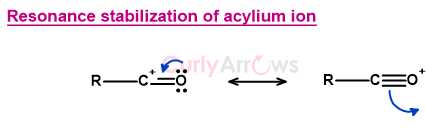
Generation of acylium ion electrophile
The first step of the reaction is the coordination of chloro of the acyl chloride with the Lewis acid AlCl3 to form a complex.

In the next step, the Oxygen lone pair delocalizes over the π bond, pushing away the coordinated chlorine-AlCl3 complex, generating the acylium ion. The acylium ion formed is stabilized by resonance.
Acylium ion-Benzene reaction
Acylium ion formed reacts with the Benzene to form the carbocation intermediate, also resonance stabilized.
(Note that substrates with electron-donating groups on the Benzene ring enable a faster acylation reaction. Also, such donor groups can stabilize the carbocation transition state by the +I effect.)
The last step is the abstraction of a proton from the carbocation intermediate by base AlCl4 to give acylated Benzene, HCl, and AlCl3 as the final products.

Here the regenerated AlCl3 is not available for further reaction as a catalyst as it coordinates with the oxygen of the carbonyl (C=O). Therefore, AlCl3 is a reagent in acylation reaction.
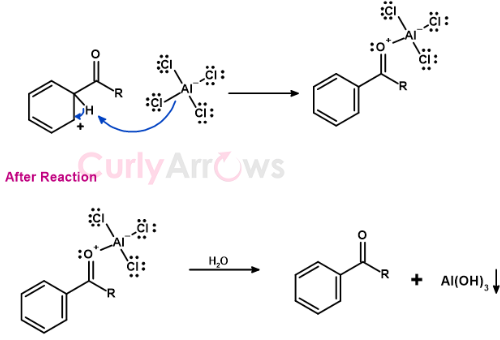
Post Reaction - Removal of Ketone
On completion of the reaction, the keto-AlCl3 product is treated with water to break the complex to generate the desired aryl ketone.
AlCl3 undergoes hydrolysis to give Aluminium hydroxide that precipitates and is removed by filtration.
