Elimination Types of Reactions in chemistry
Elimination reaction involves the loss of two groups and the formation of a new bond.
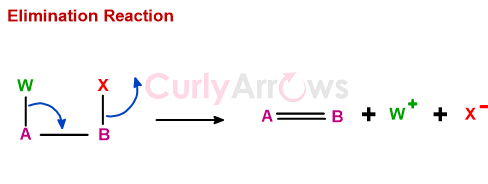
Elimination reaction takes place either by heterolytic or pericyclic mechanism.
Elimination reaction involves the loss of two groups and the formation of a new bond.

Elimination reaction takes place either by heterolytic or pericyclic mechanism.
A decomposition reaction breaks down the substrate into two or more smaller, simpler molecules.
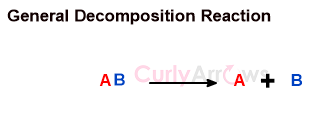
The decomposition reaction is initiated by obtaining energy from heat, light, or electricity sources.
Depending on the source, the decomposition reaction is of three types-
An atom or group of atoms migrate from one position to another; such reactions are rearrangement reactions.
Post rearrangement, the molecule formed has the atoms arranged differently but has the same molecular formula as the parent. Therefore, such rearrangements create structural isomers, compounds that differ in structure but have the same molecular formula.
Related Reading - Structural Isomerism
Two molecules combine in an addition reaction to form a single addition product (an adduct). The addition occurs in unsaturated molecules of alkenes (C=C), alkynes (C≡C), carbonyls (C=O), and imines (C=N).
There are four mechanisms in addition type of reactions-
a) Electrophilic addition (heterolytic, two electron movement)
b) Nucleophilic addition (heterolytic, two electron movement)
In the 1700s, the term radical, Latin for root, was used for any group that remained rooted or unchanged in a chemical reaction. So, the -CH3 in CH3-OH undergoing chemical transformation to CH3-Cl was radical.
Therefore, a substituent or a functional group free from a chemical transformation was called a ‘free radical.’
Only 20 naturally occurring elements exist in only one form and have no natural isotopes. Other elements occur as a mixture of two or more isotopes. The atomic weight of each isotope of an element will be different since it is the sum of the number of protons and neutrons in the atom's nucleus.
Dalton, in 1804, in his work on ‘Atomic Theory,’ proposed the law of chemical combination to explain how atoms form compounds. According to him, atoms of different elements combine in a simple whole-number ratio to give compounds.
Debye, a subtype of Vander Waal's force, is an interaction between polar-nonpolar atoms or molecules.
An example is an interaction between a polar molecule like H-Cl and a neutral, nonpolar atom like Helium.
Keesom force, a subtype of Vander Waals' is an attractive interaction occurring in polar molecules. For example, interaction between two HCl molecules.
Other examples of polar molecules are -CO2, SO2, NH3, H2O, HCl, etc.
Atoms, molecules, or ions interact by attraction or repulsion. Attraction occurs between opposite charges, and repulsion occurs when charges are the same. Therefore, most interactions are controlled by charges.