The DBE calculation uses the general molecular formula to find the presence of unsaturation in a compound. The unsaturation is calculated in levels or degrees.
The lowest degree of unsaturation (DOU) indicates minimum unsaturation, where there is the least loss of hydrogens to form a pie bond or a cyclic ring, like in cycloalkanes.
General Molecular Formula
CnH2n+2 CnH2n and CnH2n-2
saturated unsaturated
The formula for calculating DBE is -
DOU/DBE= (2C + 2 + N -H - X)/2
The calculation ignores heteroatoms like oxygen and sulfur from the molecular formula; however, it considers halogen and nitrogen.
Steps to calculate DBE
Calculate using the number of Carbon (C), Hydrogen (H), Nitrogen (N), and Halogen (X) from the molecular formula as shown below-
a) For Molecular formula C5H10 = (2C + 2 + N -H - X)/2 = (2 x 5 + 2 - 10)/2 = (12-10)/2 = 1
b) For Molecular formula C2H3OCl = (2C + 2 + N -H - X)/2 = (2 x 2 + 2 - 3 - 1)/2 = (6-4)/2 = 1
c) For Molecular formula C9H10O = (2C + 2 + N -H - X)/2 = (2 x 9 + 2 - 10)/2 = (20-10)/2 = 5
The DBE calculation does not give the exact number of the molecule’s double bond, triple bond, or ring present but offers possibilities.
For instance, a few possible combinations of the DBE values and the type of unsaturation that could be present in the compound are-
DBE Value 1 = It could come from one pie bond or from a ring in the structure
DBE Value 2 = Possible combinations:
two double bonds or
one triple bond or
two rings or
one double bond + one ring
Remember that a triple bond has two pie bonds.
DBE Value 3 = Possible combinations:
three double bonds or
one triple bond + one double bond or
three rings or
two rings + one double bond or
one ring + two double bonds
DBE value of 4 = Possible combinations:
four pie bonds or
four rings or
three pie bonds + one ring (a classic example is benzene) or
two pie bonds + two rings or
one pie bond + three rings or
two triple bonds or
one triple bond + two double bond or
one triple bond + two rings.

Example of DBE calculation from a molecule structure
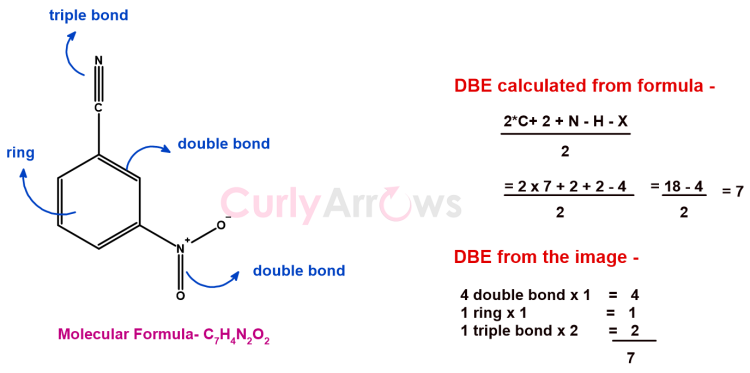
Therefore, the DBE value provides incomplete information.
More information is needed to deduce the actual structure of the molecule, which can be obtained from laboratory experiments and analytical techniques like NMR (nuclear magnetic resonance spectroscopy), IR (infrared spectroscopy), and Mass spectroscopy.
A few examples wherein the structure of the molecule is predicted from DBE value and laboratory experiments are-
Practice Problems
a) Calculate the Double Bond Unsaturation from the Molecular Formula (quiz)
b) Predict the Value of Double Bond Unsaturation from the Structure (quiz)